The endovascular aneurysm repair market size is anticipated to grow at a CAGR of 6.3% with USD 3.5 Bn in 2026 and is expected to reach USD 6.5 Bn in 2033. The growth is mainly owing to the growing demand for minimally invasive procedures alongside advancements in endovascular technologies. The increasing prevalence of aortic aneurysms (153,927 people died from aortic aneurysms across the globe), alongside the development of more efficient stent grafts and treatment solutions, is expected to drive the market’s expansion.
Fenestrated stent graft is projected to account for the largest endovascular aneurysm repair market share of 30.5% in 2026. The leadership is owing to its ability to treat complex abdominal aortic aneurysms. This is because standard EVAR grafts may not achieve a secure proximal seal, especially juxtarenal, pararenal, and short-neck anatomies. Fenestrated stent is indicated for patients with abdominal aortic or aortoiliac aneurysms and also for cases with a proximal neck length of at least 4 mm which are unsuitable for a non-fenestrated graft.
In addition, the procedural preference is growing due to improved clinical outcomes, with studies showing substantially lower morbidity and mortality rates in patients treated with fenestrated grafts compared to open surgery, thereby driving market confidence. The increasing demand for minimally invasive surgeries and advancements in catheter-based technologies are expected to fuel the growth in the market. The fenestrated stent grafts continue to demonstrate superior patient outcomes, thereby reducing complications and hospital stays.
In January 2025, Bentley launched two new products at LINC in Leipzig. The BeGraft Stent Graft System is the world’s first on-label bridging device. The BeFlared FEVAR Stent Graft System is the first dedicated bridging stent for Fenestrated EndoVascular Aneurysm Repair (FEVAR) procedures. With this launch, Bentley is taking a pioneering role in the FEVAR market.
To learn more about this report, Request Free Sample
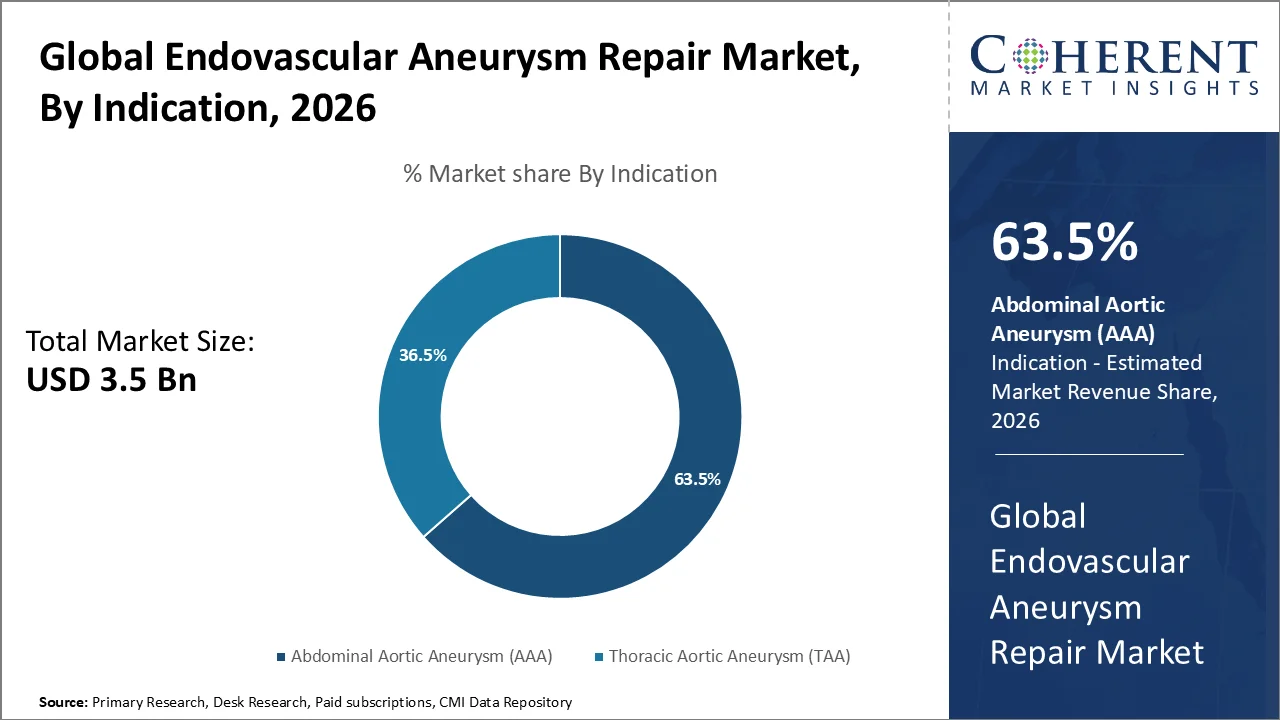
Based on indication, Abdominal Aortic Aneurysm (AAA) is poised to dominate the market and accounts for a considerable 63.5% share in 2026. Abdominal Aortic Aneurysm (AAA) is the most common type of aortic aneurysm treated via endovascular repair, specially in older individuals and those with a history of smoking. As per the data from the Centers for Disease Control and Prevention (CDC), smoking contributes to approximately 75% of AAA cases. This results in substantial expansion of the eligible patient population for screening and treatment. The growing prevalence of AAA among high-risk populations is also bolstered by aging demographics.
The recent hospital data also constitutes the safety advantages of EVAR over open surgery. Specifically, 8.01 deaths per 1,000 discharges are reported for endovascular repair of unruptured AAA, compared to higher mortality rates for open surgical repair. This safety profile has constituted to the increasing implementation of EVAR.
One such key advancement in this sector is the approval by the Centers for Medicare & Medicaid Services (CMS) of an Investigational Device Exemption (IDE) study for Terumo Aortic's Fenestrated TREO Stent-Graft System (study ID G240247/NCT07026877), which was approved in September 2025. This development is likely to propel innovation in AAA treatment and facilitate the market's growth as new, minimally invasive options become more widely accessible.
The Endovascular Aneurysm Repair (EVAR) Market in the US is undergoing substantial transformation. This is owing to the increasing shift from traditional open surgery to minimally invasive EVAR procedures. The hospital data highlights the clear safety advantages of catheter-based EVAR over open surgery. This shift is mainly driven by lower perioperative mortality, shorter hospital stays, faster recovery, reduced surgical trauma, alongside the growing preference for less invasive treatment options amongst eligible patients.
According to the AHRQ’s August 2025 IQI benchmark, using data from the 2020–2022 HCUP State Inpatient Databases, 57.06 deaths per 1,000 discharges occurred for open repair of unruptured abdominal aortic aneurysms (AAA). Whereas only 8.01 deaths per 1,000 discharges were reported for EVAR. This substantial reduction in mortality is propelling greater physician and hospital preference for EVAR, particularly for eligible patients.
In addition, the recent regulatory advancements are further speeding up the EVAR adoption. For instance, the Centers for Medicare & Medicaid Services (CMS) approved the IDE study G240247/NCT07026877 for Terumo Aortic’s Fenestrated TREO Stent-Graft System on September 2025. This marks a key milestone in EVAR technology development.
The development of fenestrated and branched stent grafts is substantially advancing the Endovascular Aneurysm Repair (EVAR) market by extending treatment options to patients with complex thoracoabdominal and pararenal aneurysms.
In January 2024, the U.S. FDA approved the GORE EXCLUDER Thoracoabdominal Branch Endoprosthesis (TAMBE) for endovascular repair in patients with thoracoabdominal aortic aneurysms and high-surgical-risk pararenal aneurysms who have suitable anatomy. This stent graft uses four portals to maintain blood flow to vital arteries, including the SMA, celiac artery, and renal arteries.
Additionally, in January 2025, the FDA cleared a supplement for the reduced-profile GORE VIABAHN VBX. It is now approved as a branch component for TAMBE, thereby facilitating lower-profile branch access. A post-approval study involving 300 adults in 60 United States sites is underway to confirm the real-world benefit-risk of this device.
|
Current Event |
Description and its Impact |
|
Regulatory Updates on Medical Device Approval Processes in the EU and U.S. (2026) |
|
|
U.S. Medicare Reimbursement Policy Adjustments for Endovascular Aneurysm Repair Procedures (2026) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
To learn more about this report, Request Free Sample
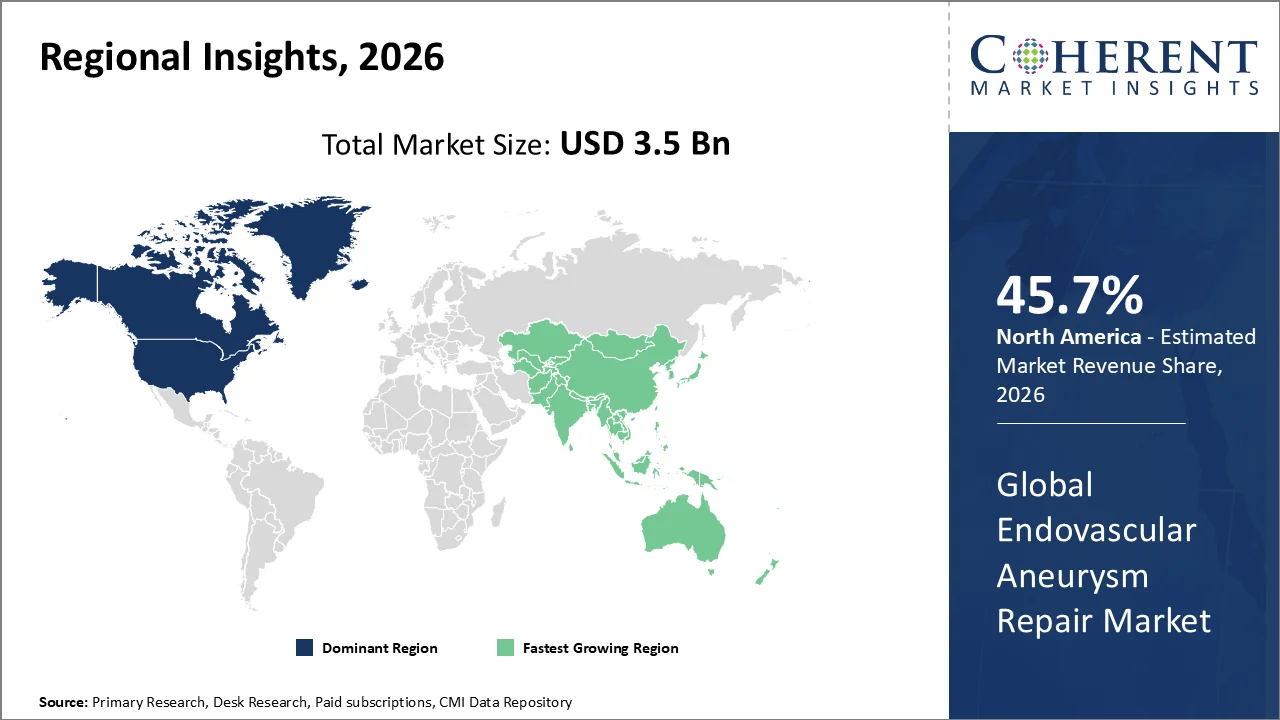
North America is set to account for the leading position in the endovascular aneurysm repair market. The region occupies a substantial 45.7% share in 2026. The growth is mainly driven by the strong presence of advanced vascular surgery centers, high adoption of minimally invasive aneurysm repair techniques, and wider availability of trained interventional radiologists and vascular surgeons.
Hospitals across the Canada and United States are preferring EVAR over open surgical repair for eligible patients because it facilitates shorter hospital stays, lower procedural trauma, faster recovery, and improved suitability for elderly or high-risk patients. The region also benefits from early access to advanced stent graft systems, fenestrated grafts, branched grafts, and image-guided endovascular platforms.
Favorable reimbursement for medically necessary aneurysm repair, strong clinical trial activity, and continuous product innovation by leading medical device companies further strengthen regional adoption.
The Asia Pacific region is poised to be as the fastest-growing region through 2026-2033. The region’s growth is owing to a rising procedure volume, aging populations, and the increasing adoption of minimally invasive techniques.
The growth is particularly significant in countries like Japan, where a 3.8% increase in endovascular aneurysm repair procedures was reported in 2025. This included 5,902 cases of non-dissected aortic aneurysms and 4,090 TEVAR procedures, thus highlighting a major shift toward stent-graft technology. The peripheral arterial diseases like abdominal aortic aneurysms were responsible for 25% of all deaths in the country of Australia in 2022. This statistic reflects the urgent need for both improved screening and treatment alternatives.
One of the major developments in the region was the release of Terumo India’s TREO Abdominal Stent-Graft System in August 2024. It was mainly designed for infrarenal aneurysm repair and ensured greater access to cutting edge EVAR treatments, particularly in underserved areas. TREO is positioned as an advanced EVAR graft with both suprarenal and infrarenal active fixation. As healthcare infrastructure improves, the market is expected to expand rapidly, driven by both technological innovations and increased awareness about aortic diseases.
The United States Endovascular Aneurysm Repair Market is witnessing robust activity mainly driven by increased adoption of minimally invasive techniques. The recent U.S. data indicate that approximately 80% of abdominal aortic aneurysm (AAA) repairs are now performed using EVAR, thus reflecting widespread substitution of open surgery with endovascular methods. The use of Intact AAA EVAR has increased from 45% to 66% over recent years, while open repair rates have decreased. These trends demonstrate an ongoing shift toward less invasive care that reduces perioperative risk.
One of the key development in October 2025 was the FDA labeling approval update for Medtronic’s Endurant stent graft system which allowed treatment of ruptured abdominal aortic aneurysm (rAAA). This approval is expected to further drive EVAR adoption, enhancing physician confidence and broadening its use in emergent EVAR procedures.
The China Endovascular Aneurysm Repair Market is poised for steady growth over the forthcoming years. The growth is owing to a rapidly ageing population and advancements in domestic stent graft approvals. According to the National Bureau of Statistics of China, in 2025, approximately 323 million people were aged 60 and above, making up about 23% of the total population. These growing elderly demographic is one of the main drivers for the increased demand for minimally invasive aneurysm repair procedures like EVAR.
One such example of regulatory developments is the China National Medical Products Administration (NMPA) officially approving the G‑iliac™ Pro Iliac Artery Stent Graft System on January 2026. It was designed for endovascular treatment of abdominal aortic and iliac artery aneurysms, thereby enhancing available options in EVAR therapies.
Additionally, ongoing clinical studies registered on the Chinese Clinical Trial Register are evaluating innovative triple‑branch aortic stent‑graft systems for endovascular repair, indicating continued clinical development activity in this market segment.
Some of the major key players in Endovascular Aneurysm Repair Industry are Medtronic plc. Cook Medical, Gore Medical, B. Braun Melsungen AG, Terumo Corporation, Boston Scientific Corporation, Abbott Laboratories, Volcano Corporation, C.R. Bard, Spectranetics International BV, Cordis Corporation (Johnson and Johnson Co.), and St. Jude Medical Inc.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 3.5 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: |
6.3% |
2033 Value Projection: | USD 6.5 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Medtronic plc. Cook Medical, Gore Medical, B. Braun Melsungen AG, Terumo Corporation, Boston Scientific Corporation, Abbott Laboratories, Volcano Corporation, C.R. Bard, Spectranetics International BV, Cordis Corporation (Johnson and Johnson Co.), and St. Jude Medical Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients