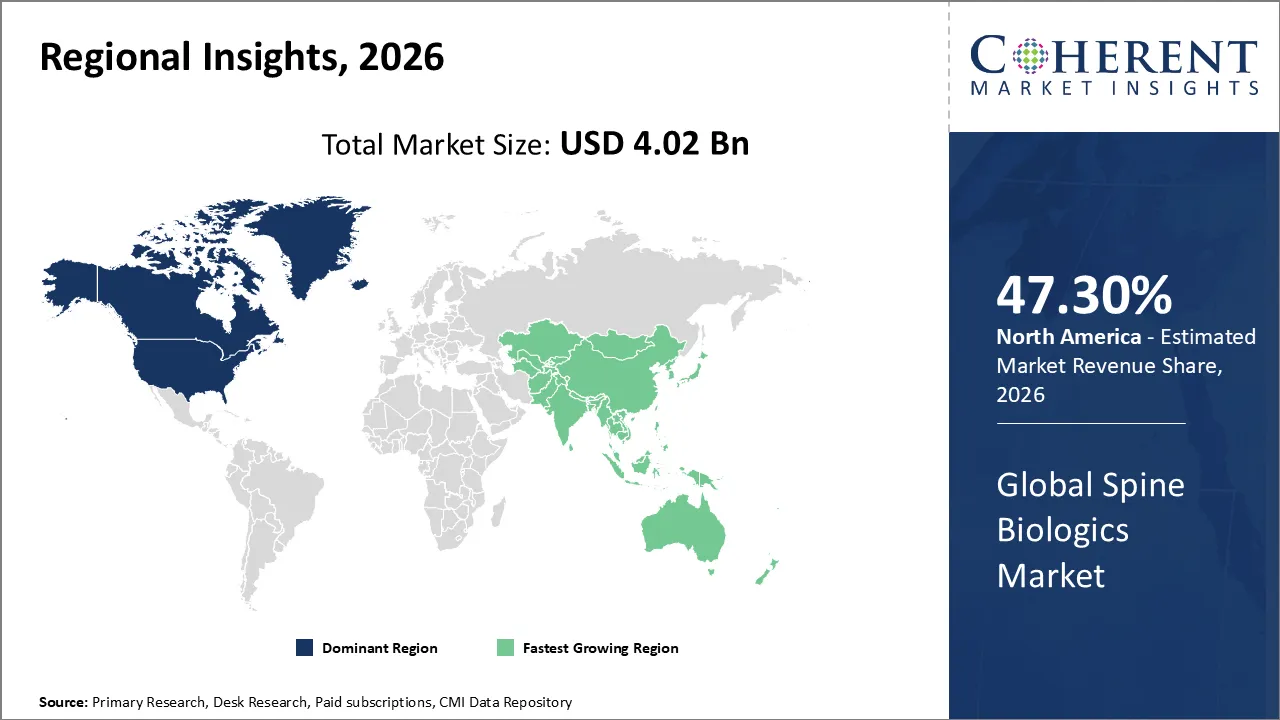
The spine biologics market is estimated to be valued at USD 4.02 Bn in 2026 and is expected to reach USD 5.70 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 5.15% from 2026 to 2033.
The global spine biologics market is expanding steadily, driven by rising spinal disorders and demand for regenerative treatments. Growth is supported by increasing cases of degenerative disc disease, advancements in biologics such as stem cells and growth factors, and strong adoption across North America and Asia-Pacific. Innovation in minimally invasive procedures and heightened awareness of biologic therapies are expected to further accelerate market penetration over the coming years.
|
Current Event |
Description and its Impact |
|
Regulatory Evolution and FDA Policy Changes |
|
|
Technological Disruption and Innovation Breakthroughs |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
In 2026, policies regarding how to pay for spine biologics are an important factor why people want them in the U.S. and China. Medicare and private insurance companies in the US are covering increasingly sophisticated biologics like BMPs, DBM, and cell-based matrices, especially in minimally invasive fusion procedures. Bundled payment models also lower hospital costs and make it easier for people to get care. The National Reimbursement Drug List in China now includes biologics, and the government gives funding to help people pay for them. This means that China does not rely as much on imports and biologics are more economical. These good reimbursement frameworks work together to make it easier for patients to get care, encourage hospitals to adopt them, and speed up market growth in both regions.
In terms of product type, the bone graft substitute segment is expected to lead the market with 47.6% share in 2026, due to their versatility and reduced complications compared to autografts. Surgeons now have safer, more accessible options due to allografts, demineralized bone matrix (DBM), synthetics, and xenografts. Since they can speed up fusion rates while lowering the risk of complications at the donor site, they will be widely used in spinal surgeries.
For instance, in October 2025, the launch of Aurora Spine's new biologics portfolio demonstrates the way new ideas are being used in bone graft substitutes. TURBO FUSE™ Fiber Putty, OSTEO GRAFT™ DBM Putty, and OSTEO BRIDGE™ Sponge Strips are all safer, allograft-based products that improve the results of spinal fusion.
In terms of surgery type, the Posterior Lumbar Interbody Fusion (PLIF) segment is expected to hold 32.8% share in 2026, due to its proven stability and effectiveness in treating degenerative disc disease. Surgeons favor PLIF as it works well, helps patients get better, and works well with biologics that speed up bone healing. Its widespread use solidifies its position as the best method for spinal fusion.
For instance, in February 2024, Amplify Surgical's highlights its dualX® expandable interbody devices, which are made for lumbar fusion surgeries like Posterior Lumbar Interbody Fusion (PLIF). When used with dualPortal® endoscopic techniques, these new ideas make implant expansion more accurate, bone grafting better, and fusion outcomes better. This strengthens Amplify's role in advancing minimally invasive spine biologics solutions.
In terms of end user, the hospital segment is projected to account for 57.3% share in 2026, due to advanced infrastructure, skilled surgeons, and capacity to handle complex spinal surgeries. They get the most patients, which lets them use the newest biologics. In 2026, hospitals are still the main place where biologic-based spinal fusion treatments are done, even though orthopedic clinics and ambulatory centers are growing steadily.
For instance, in May 2025, Xtant Medical announced that OsteoFactor® PRO, a new biologic that is supposed to improve bone growth and spinal fusion outcomes, is now available. This product is meant for spinal surgeries in hospitals. It adds to Xtant's line of biologics and gives surgeons more advanced and effective options that increase the chances of fusion success and make it easier for doctors to use in complex spine surgeries.
To learn more about this report, Download Free Sample
North America is dominating the spine biologics market with 47.3% share in 2026, due to advanced healthcare infrastructure, high prevalence of spinal disorders, and strong adoption of bone graft substitutes. Major players like Medtronic and Stryker drive innovation, while favorable reimbursement policies and hospital expertise ensure rising demand for biologics in spinal fusion procedures.
For instance, in November 2025, Xtant Medical released CollagenX™ in the US, adding to its line of biologics for the spine. CollagenX is a biologic made from collagen that helps bones heal and fuse together after spinal surgery. This commercial launch gives Xtant a stronger foothold in the North American spine market and sets the company up for future chances to grow globally.
Asia Pacific is anticipated to be the fastest growing region, due to its large patient pool, rising spinal disorder prevalence, and rapidly aging population. Expanding healthcare infrastructure, increasing investments, and growing adoption of minimally invasive fusion techniques in countries like China, India, and Japan fuel the region’s fastest growth.
For instance, in August 2025, VB Spine grew around the world when it acquired Stryker's spinal implants business in Australia and New Zealand. This strategic move strengthens VB Spine's portfolio in spinal fixation technologies and increases its presence in the Asia-Pacific spine surgery market, putting the company in a good position to grow in one of the fastest-growing areas for advanced spinal solutions.
The U.S. spine biologics market in 2026 is driven by rising cases of degenerative disc disease, spinal injuries, and scoliosis, alongside growing adoption of advanced biologics like DBM, BMPs, and cell-based matrices. FDA approvals, technological innovations, and surgeon preference for minimally invasive fusion procedures fuel strong demand and market expansion.
For instance, in July 2025, Orthofix introduced the Reef-L Interbody System to the US for lateral lumbar fusion surgeries. Reef-L uses 3D-printed titanium technology to improve bone growth and stability in spine surgeries that not require an excessive amount of surgery. This commercial launch helps Orthofix's presence in the U.S. in spinal implants and biologics, giving surgeons better fusion solutions.
The spine biologics market in China in 2026 is driven by rising cases of degenerative spine disorders, trauma, and aging population needs, alongside increasing adoption of minimally invasive fusion procedures. Government healthcare reforms, expanding hospital infrastructure, and surgeon preference for advanced biologics like BMPs and DBM fuel strong demand and market growth.
For instance, in December 2025, SpineGuard obtained regulatory approval in China for three new PediGuard models, which adds to its line of smart drilling tools. PediGuard lowers the risks of surgery by measuring electrical conductivity in real time, which makes it easier to place pedicle screws correctly. This approval makes SpineGuard's presence in the Asia-Pacific spine surgery market stronger, adding to its existing presence in the U.S. and Europe.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 4.02 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.15% | 2033 Value Projection: | USD 5.70 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Fuse Medical Inc., Medtronic plc., Zimmer Biomet, Wright Medical Group Inc., NuVasive Inc., Stryker Corporation, EmCyte Corporation, OrthoPediatrics Corp., Baxter International, HemaCare Corporation, K2M Inc., Adimarket, LLC, and Orthofix Medical Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The increasing prevalence of spinal disorders, including degenerative disc disease, spinal stenosis, and trauma-related injuries, is a major growth driver for the spine biologics market size. Rising cases among aging populations and younger individuals with sedentary lifestyles fuel demand for biologics, enhancing fusion success rates and expanding market adoption.
Advancements in biologic technologies significantly boost the spine biologics market share by improving fusion outcomes and reducing complications. New treatments like stem cell therapies, platelet-rich plasma (PRP), bone marrow aspirate concentrate (BMAC), and synthetic grafts make it easier for the body to heal itself. These breakthroughs make surgeons prefer them, patients use them, and the market keep growing around the world.
The expansion of regenerative therapies, including stem cells, platelet-rich plasma (PRP), and bone marrow aspirate concentrate (BMAC), is a major growth catalyst in the spine biologics market forecast. These advanced biologics enhance spinal fusion success, accelerate healing, and reduce complications, driving strong adoption and shaping long-term market opportunities globally.
Innovation in synthetic bone grafts is driving higher spine biologics market demand by offering safer, readily available alternatives to autografts. Advanced biomaterials, demineralized bone matrix (DBM), and bioactive synthetics reduce donor-site morbidity while enhancing fusion success. These breakthroughs improve patient outcomes, increase surgeon preference, and accelerate adoption across global markets.
The spine biologics market value is growing steadily as more individuals are getting spinal disorders, more biologic materials are being used in spinal fusion and repair procedures, and new regenerative therapies are always being developed. Estimates for the global market say that the industry is expected to be worth over 2.5 billion in 2025. Projections show that it will keep growing for the next ten years as more doctors accept biologic solutions. Allografts and substitutes are two types of bone graft materials that are still very important to the market. This is as they help bones heal faster, increase the chances of fusion success, and lower the need for traditional autografts.
Clinical utilization patterns indicate that spinal allografts rank among the most extensively employed biological products, particularly in fusion surgeries such as anterior cervical discectomy and posterior lumbar interbody fusion. Cell-based matrices and platelet-rich plasma are becoming more popular as they can help the body heal and as more people are interested in less invasive methods.
Hospitals and specialized spine surgery centers use the most spine biologics. This is since they have structured surgical workflows and multidisciplinary teams that can incorporate advanced biologic products into treatment plans. North America has the highest regional consumption due to the fact has a well-established healthcare system, surgeons prefer biologic solutions, and there are more procedures. As healthcare systems get better and access to advanced spinal care gets better, Asia-Pacific is growing the fastest.
The market is growing, but it continues to face problems. For example, there are high regulatory barriers for new biologic materials and different reimbursement frameworks across healthcare systems. These can affect the speed at which people adopt products and how prices change.
Share
Share
Nikhilesh Ravindra Patel is a Senior Consultant with over 8 years of consulting experience. He excels in market estimations, market insights, and identifying trends and opportunities. His deep understanding of the market dynamics and ability to pinpoint growth areas make him an invaluable asset in guiding clients toward informed business decisions. He plays a instrumental role in providing market intelligence, business intelligence, and competitive intelligence services through the reports.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients