The Vaccine Adjuvant market is anticipated to grow at a CAGR of 2.34% with USD 1.81 Bn in 2026 and is expected to reach USD 2.13 Bn in 2033. The global vaccine adjuvant market is experiencing strong growth due to the rise in prevalence of infectious diseases (States documented 23,613 confirmed cases, and Canada reported 11,646 cases) and increasing usage of adjuvants in vaccines worldwide. Moreover, increasing government funding for vaccine research, rising adoption of traditional vaccines, development of oil-in-water and water-in-oil emulsions, technological advancements in pathogen component adjuvants are the factors responsible for the growth of the market.
Infectious disease dominates the market, accounting for a significant 70% share in 2026, the fundamental role vaccines play in global public health initiatives is one of the factors responsible for the growth of the segment. The continuous emergence of infectious disease threats worldwide. In 2025, 184 countries and areas with more than 99% of the world’s population and TB cases reported data.
The World Health Organization's Expanded Programme on Immunization has been instrumental in driving demand for adjuvanted vaccines against infectious diseases. For example, the widespread use of aluminum-based adjuvants in vaccines against hepatitis B, diphtheria, tetanus, as well as pertussis has proved the importance of adjuvants in improving immune responses against infectious pathogens.
Mineral Salt-based Adjuvant is projected to account for the largest share type in 2026, representing approximately 40% of the total volume. Their extensive regulatory approval history, proven safety profile, as well as widespread adoption across multiple vaccine platforms are the factors responsible for the growth of the segment. Immunization currently prevents 3.5 million to 5 million deaths every year from diseases like diphtheria, tetanus, pertussis (whooping cough), influenza, etc.
The dominance of mineral salt-based adjuvants, particularly aluminum-based compounds such as aluminum hydroxide, aluminum phosphate, as well as aluminum potassium sulfate, dates back from their pioneering role in vaccine development back in 1920s.
The World Health Organization has consistently endorsed aluminum adjuvants as safe as well as effective components in human vaccines, with regulatory bodies like the FDA and EMA having approved a number of vaccines containing these adjuvants. Major pharmaceutical companies including GlaxoSmithKline, Merck, Pfizer, Sanofi, etc., have incorporated aluminum adjuvants in their widely distributed vaccines such as hepatitis A and B vaccines, diphtheria-tetanus-pertussis combinations, as well as human papillomavirus vaccines.
Active Immunostimulants dominates the market, accounting for a significant 50% share in 2026, their critical role in enhancing immune responses as well as their widespread adoption across various vaccine formulations is bolstering the growth of the segment. Active immunostimulants operate by directly stimulating the response mechanisms of immune system, making them indispensable components in modern vaccine development.
The World Health Organization has consistently targeted the importance of adjuvants that can provoke robust immune responses, particularly in populations with compromised immune systems such as elderly individuals as well as immunocompromised patients.
Active immunostimulants are popular as they work well, needing less antigen while making the immune response stronger as well as last longer.
To learn more about this report, Request Free Sample
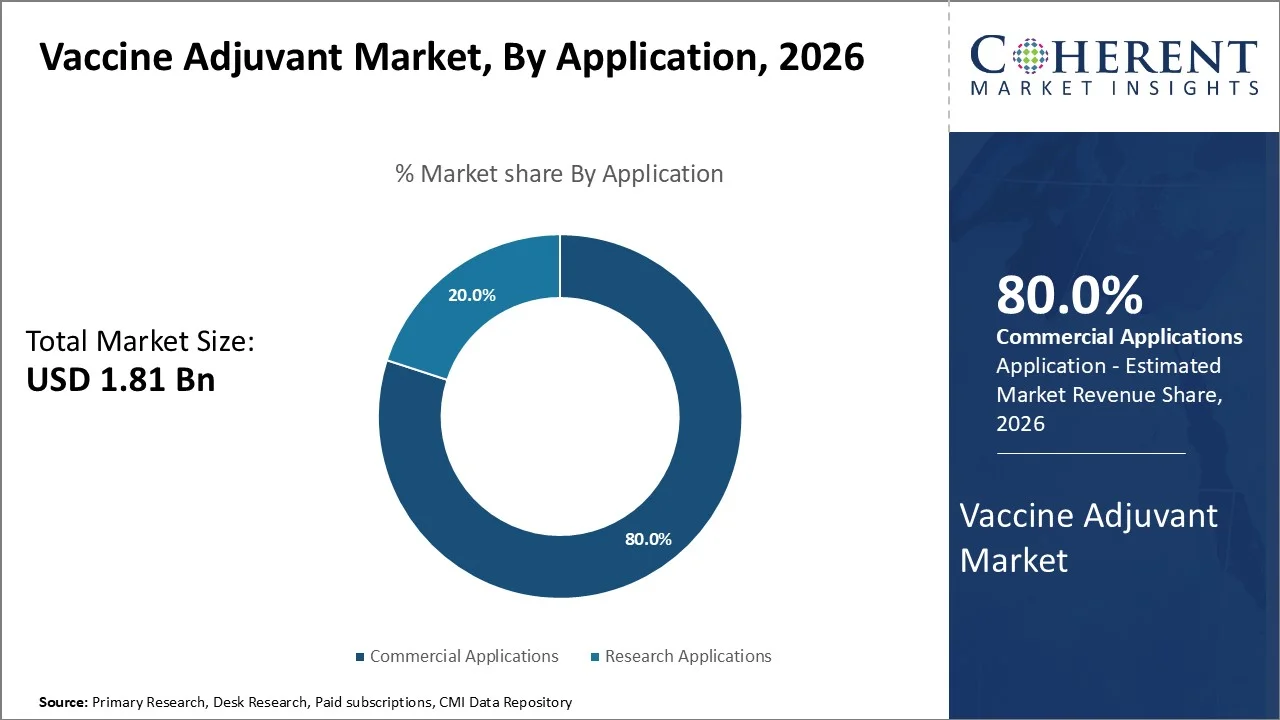
Commercial applications dominate the market, accounting for a significant 80% share in 2026, owing to the widespread deployment of adjuvanted vaccines in routine immunization programs worldwide and the substantial investment by pharmaceutical companies in developing commercially viable vaccine formulations.
Bacterial meningitis is an often-deadly infection which also leaves 1 in 5 individuals with long-term devastating sequelae after the acute phase. By end 2024, 24 of 26 countries in the belt conducted preventive campaigns, and 15 introduced MenAfriVac in their routine immunization programme, reaching 410 million people. Coverage is estimated at 60% in countries using MenAfriVac in routine immunization programmes and 29% overall in the 26 countries.
The commercial segment includes vaccines approved by agencies like the FDA, EMA, and WHO, and sold through healthcare systems worldwide. For example, GlaxoSmithKline’s AS04 booster, which has aluminum hydroxide and monophosphoryl lipid A, is used in vaccines like Cervarix for HPV and Fendrix for hepatitis B in dialysis patients.
New adjuvant technologies use nanotechnology, like lipid nanoparticles (LNPs), to deliver vaccines. These tiny particles carry both the vaccine and immune-boosting molecules, release them slowly, as well as aid target important immune cells called dendritic cells.
New liposomal vaccines are stronger as well as stay effective at different temperatures. This aid with storing as well as moving vaccines, especially in countries with fewer resources.
|
Current Event |
Description and its Impact |
|
FDA Accelerates Approval of Novel Vaccine Adjuvants |
|
|
GAVI and WHO Vaccine Programs in Developing Countries |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
To learn more about this report, Request Free Sample
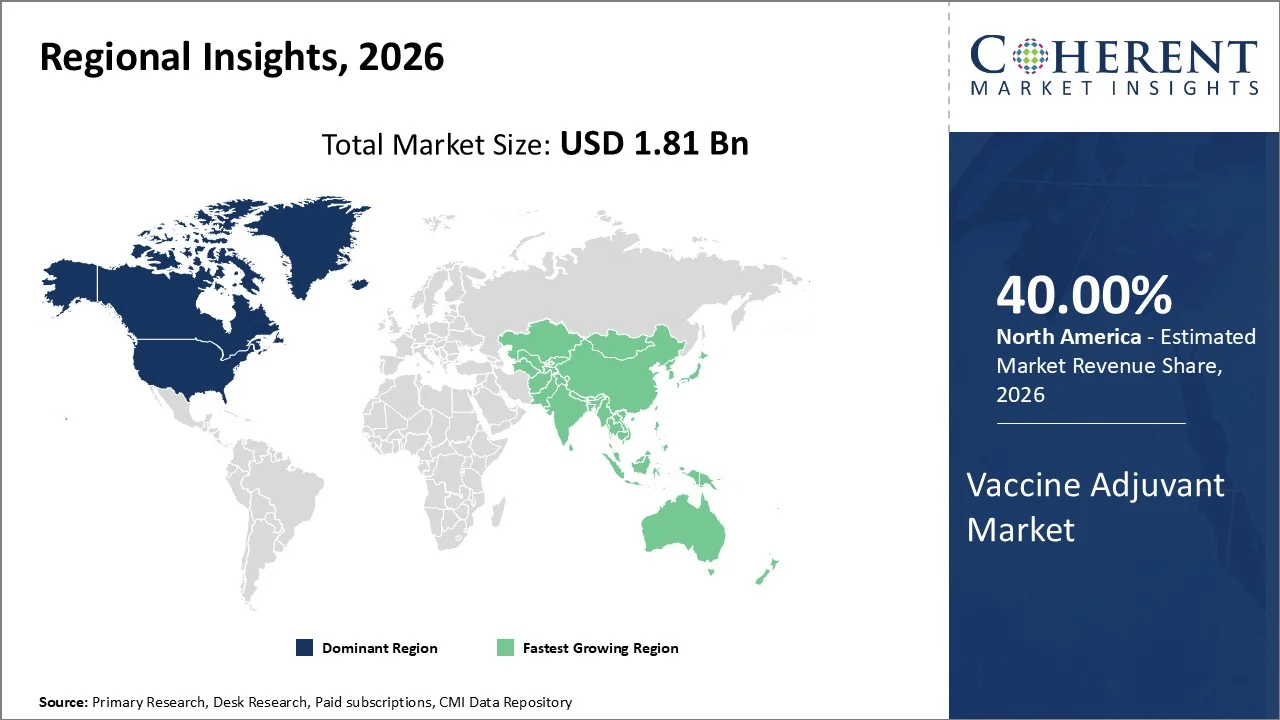
North America account 40% market share in 2026, owing to its robust pharmaceutical infrastructure, extensive research and development capabilities, and well-established regulatory frameworks that facilitate vaccine adjuvant innovation and commercialization. PhRMA member companies are delivering USD 500 billion in new U.S.-based manufacturing and infrastructure investment, injecting an estimated USD 1.2 trillion into the economy.
In January 2026, PhRMA built a search engine that point patients to prescription medicine direct purchase programs (DPPs), independently set up by biopharmaceutical manufacturing companies. AmericasMedicines.com will connect patients to DPPs where they can buy prescriptions directly from manufacturers as well as search the site’s 750+ patient assistance resources.
The region's dominance stems from the presence of major pharmaceutical corporations such as GlaxoSmithKline, which operates significant research facilities in the United States and has developed advanced adjuvant systems like AS04 used in HPV vaccines.
The Asia-Pacific region is poised to be the fastest-growing region through 2026-2033, expanding at a CAGR of approximately 7.5%. The unprecedented initiatives by government authorities along with rapidly expanding indigenous pharmaceutical capabilities across key markets are inducing growth in countries including China, India, as well as Southeast Asian nations. South Asia achieved highest-ever immunization coverage for children, with notable progress in India, Nepal, and Pakistan.
National Medical Products Administration of China approving a number of domestic vaccine adjuvant formulations, including novel aluminum-based and oil-in-water emulsion adjuvants developed by companies like Sinovac and CanSino Biologics is further bolstering the growth of the market.
The Serum Institute of India has significantly expanded its adjuvant production capabilities, partnering with local biotechnology firms to introduce cost-effective adjuvant systems that cater to tropical disease vaccines prevalent in the region.
The U.S. contributes the highest share in the vaccine adjuvant market in North America due to its robust pharmaceutical infrastructure, extensive research as well as development capabilities, and presence of major biotechnology companies. The United States houses leading pharmaceutical giants such as Pfizer, Moderna, Johnson & Johnson, etc., which have significantly invested in adjuvant research and development.
The FDA’s fast approval and emergency rules have helped vaccine booster technologies develop quickly.
China contributes highest share in market in Asia Pacific due to its robust pharmaceutical manufacturing infrastructure, extensive investment by government authorities in vaccine development, as well as established regulatory framework. This has made the country as the regional leader in vaccine adjuvant production and innovation.
The country's pharmaceutical giants, including Sinovac Biotech, Sinopharm, CanSino Biologics, etc., have demonstrated exceptional capabilities in vaccine adjuvant utilization, particularly evident during the COVID-19 pandemic when Chinese manufacturers produced billions of doses using various adjuvant technologies.
Some of the major key players in vaccine adjuvant market include, Thermo Fisher Scientific Inc., Air Liquide (Seppic), Pacific GeneTech Limited, OZ Biosciences, Novavax Inc., Merck & Co. Inc., InvivoGen, GlaxoSmithKline PLC, Dynavax Technologies Corporation, CSL Limited, Croda International PLC, Vertellus, Aphios Corporation, Agenus Inc., Adjuvatis, and Associated British Foods PLC (SPI Pharma Inc.), among others.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1.81 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 2.34% | 2033 Value Projection: | USD 2.13 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Thermo Fisher Scientific Inc., Air Liquide (Seppic), Pacific GeneTech Limited, OZ Biosciences, Novavax Inc., Merck & Co. Inc., InvivoGen, GlaxoSmithKline PLC, Dynavax Technologies Corporation, CSL Limited, Croda International PLC, Vertellus, Aphios Corporation, Agenus Inc., Adjuvatis, and Associated British Foods PLC (SPI Pharma Inc.), among others. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients