Global Alpha-1 Antitrypsin Deficiency Augmentation Therapy Market Size and Forecast – 2026 To 2033
The global Alpha-1 antitrypsin deficiency augmentation therapy market is expected to grow from USD 1,878.2 Mn in 2026 to USD 2,461.9 Mn by 2033, registering a compound annual growth rate (CAGR) of 3.94%. Rising burden of respiratory diseases linked to Alpha-1 Antitrypsin Deficiency (AATD) drives the market growth.
According to the Lancet publication, Alpha-1 Antitrypsin Deficiency (AATD) is estimated to affect about 1 in 2,000 to 1 in 5,000 people globally, and it is among the most prevalent inherited respiratory diseases.
(Source: The Lancet)
Key Takeaways of the Global Alpha-1 Antitrypsin Deficiency Augmentation Therapy Market
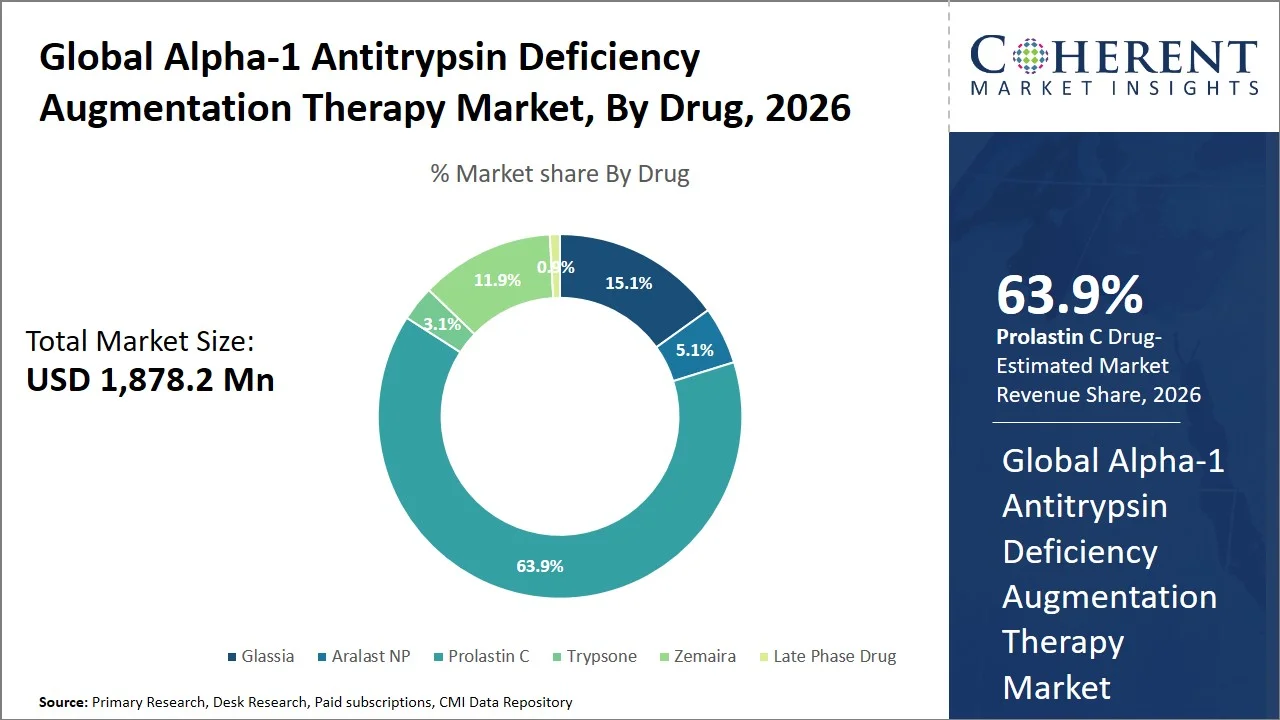
- Prolastin C is projected to hold 63.9% of the global Alpha-1 antitrypsin deficiency augmentation therapy market share in 2026, making it the dominant drug segment, especially in North America, where there is an extensive network for plasma collection, diagnostic testing programs, and favorable reimbursement systems (e.g., Medicare Part B, Medicaid, private insurers, orphan drug reimbursement frameworks) that make it widely used.
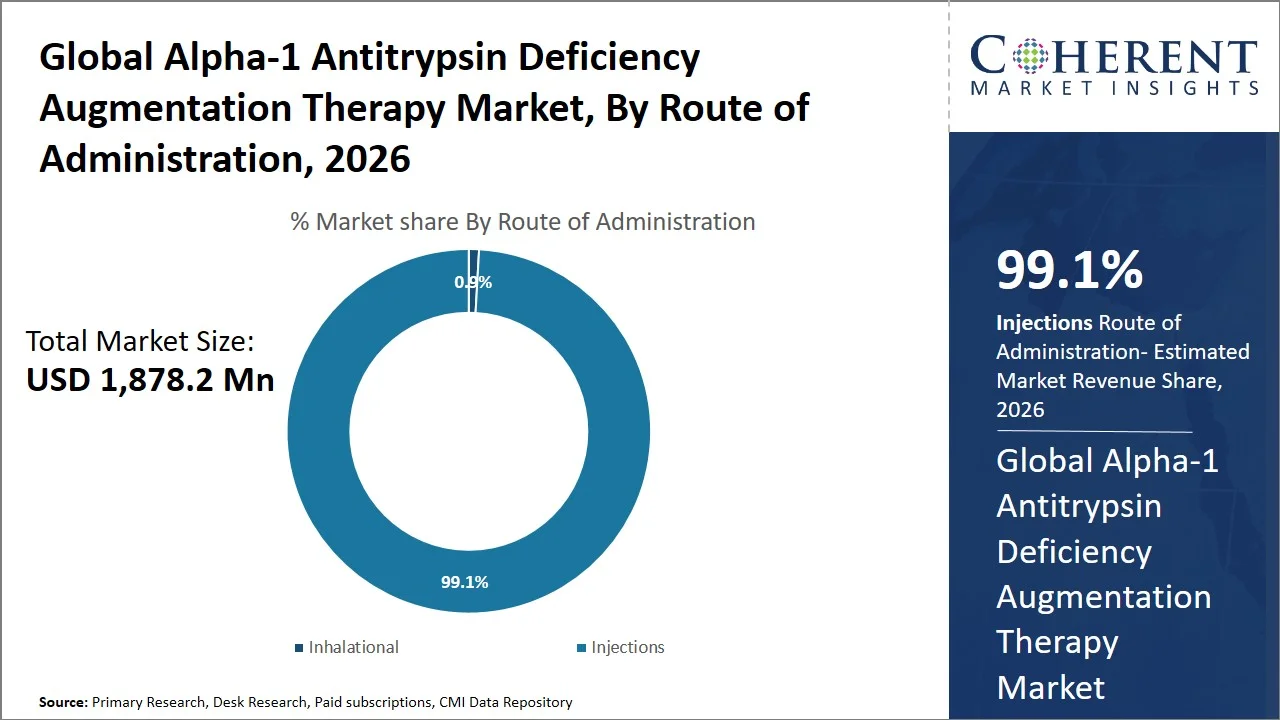
- Injections are projected to hold 99.1% of the global Alpha-1 antitrypsin deficiency augmentation therapy market share in 2026, making it the dominant route of administration segment, particularly in Europe and North America, because of the regulatory approval of only the intravenous form of the medication by organizations such as the U.S. Food and Drug Administration and the requirement in clinical guidelines for direct injection into the bloodstream for effective levels of Alpha-1 Antitrypsin (AAT) in the serum. For example, therapies approved by the U.S. FDA, such as Prolastin-C and Glassia, are only given through weekly intravenous infusions. This highlights the importance of injections delivery for effective disease management.
- Hospital Pharmacies segment is projected to hold 61.9% of the global Alpha-1 antitrypsin deficiency augmentation therapy market share in 2026, making it the dominant distribution channel segment, especially in well-developed areas such as North America and Europe, because of modern facilities in hospitals, easy availability of infusion centers, and good reimbursement system including Medicare Part B coverage, Medicaid coverage, Diagnosis Related Group (DRG) payments, and national health care systems.
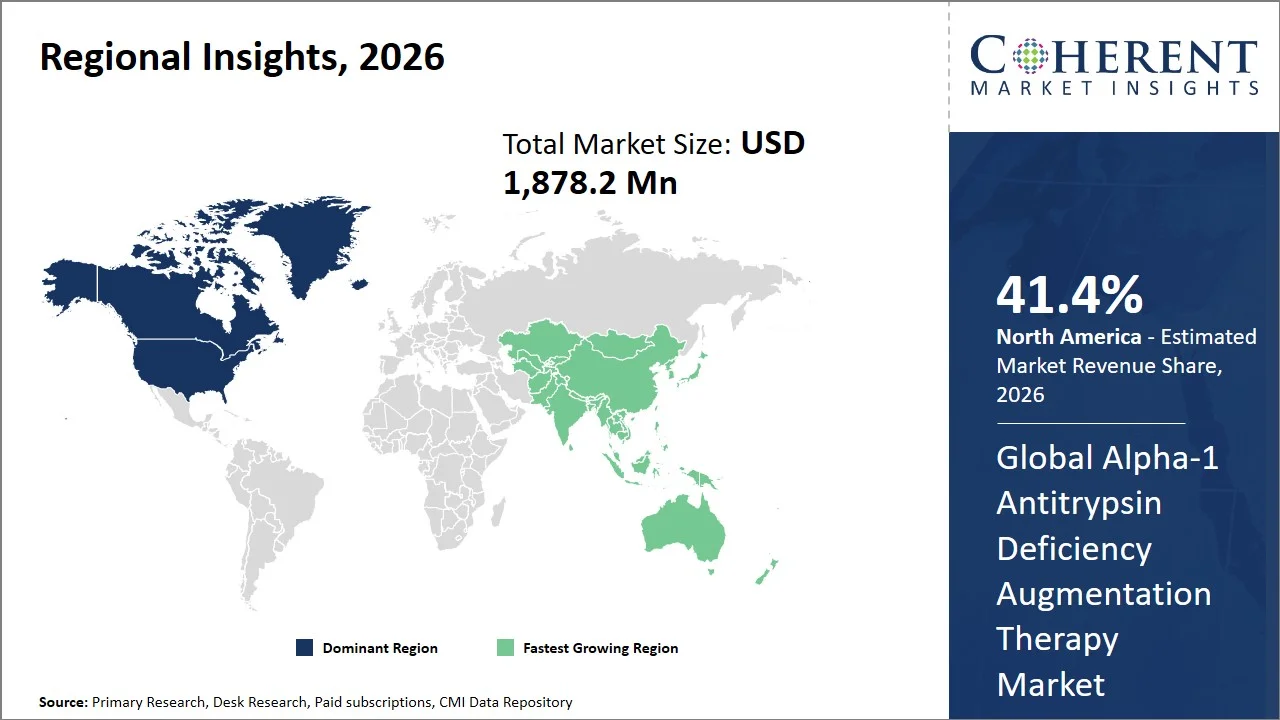
- North America market maintains dominance with an expected share of 41.4% in 2026, due to higher diagnostic frequency for Alpha-1 Antitrypsin Deficiency (AATD), availability of effective treatments like Prolastin-C, supportive reimbursement programs, and sophisticated technology in collecting and infusing plasma.
- Asia Pacific is expected to exhibit the fastest growth with an estimated contribution of 20.4% share in 2026, driven by advancements in the healthcare infrastructure, increased awareness and detection of rare disease cases, availability of biologic drugs, and increased government expenditure on healthcare facilities in countries like China and India (e.g., China spends ~5.5% of GDP on healthcare, while India spends ~3.3–3.8% of GDP).
- Expansion of recombinant Alpha-1 Antitrypsin (AAT) therapies: The transition from plasma-derived to recombinant AAT therapies presents a burgeoning growth opportunity, stemming from a decreased reliance on constrained plasma resources and the facilitation of scalable production methods. This evolution is anticipated to enhance treatment accessibility and foster sustained market expansion.
- Increasing genetic screening and early diagnosis initiatives: The increasing utilization of genetic testing and targeted screening initiatives is facilitating the earlier detection of Alpha-1 Antitrypsin Deficiency (AATD), especially within the Chronic Obstructive Pulmonary Disease (COPD) patient demographic. Consequently, this is broadening the diagnosed patient population and accelerating the commencement of augmentation therapy.
Why Does Prolastin C Dominate the Global Alpha-1 Antitrypsin Deficiency Augmentation Therapy Market?
Prolastin C is projected to hold the market share of 63.9% in 2026, due to its solid regulatory approval, lack of competition, and effective treatment validated by the data from governmental institutions. The drug has been approved by the U.S. FDA for use in chronic augmentation therapy in adults with Alpha-1 Antitrypsin Deficiency (AATD), being one of a few approved drugs from plasma and making competition in the market limited (Source: U.S. Food and Drug Administration). Moreover, clinical studies cited by the National Library of Medicine prove that Alpha-1 Antitrypsin (A1AT) augmentation therapy helps slow down the deterioration of lung functions in emphysema patients (Source: National Library of Medicine). Along with strong financial incentives for reimbursement that the developed healthcare system provides, these attributes make Prolastin C the global market leader.
Why are Injections the Most Preferred Route of Administration in Global Alpha-1 Antitrypsin Deficiency Augmentation Therapy Market?
To learn more about this report, Request Free Sample
Injections are projected to hold a market share of 99.1% in 2026. This preference stems from their capacity to facilitate the direct introduction of functional Alpha-1 Antitrypsin (AAT) protein into the circulatory system, thereby facilitating the swift attainment of protective serum concentrations. Prominent therapies, including Prolastin-C and Zemaira, are delivered via IV infusion, given that Alpha-1 Antitrypsin (AAT), being a sizable protein, would undergo degradation if administered orally. The U.S. Food and Drug Administration has sanctioned only intravenous Alpha-1 Antitrypsin (AAT) therapies, thereby solidifying injections as the established standard of care. In addition, the National Institutes of Health (NIH) recognizes the need for maintaining adequate levels of circulating Alpha-1 Antitrypsin (AAT), which can be best done by administering Alpha-1 Antitrypsin (AAT) through intravenous injection because intravenous augmentation has been found to raise AAT levels in the blood. (Source: National Library of Medicine)
Hospital Pharmacies Segment Dominates the Global Alpha-1 Antitrypsin Deficiency Augmentation Therapy Market
The hospital pharmacies segment is projected to hold a market share of 61.9% in 2026, owing to the requirements associated with the regulated delivery of the intravenous biologics and specialized monitoring required. Augmentation therapies such as Prolastin-C and Zemaira have to be administered through IV, often starting from a hospital environment to handle the possible side effects. Such drugs are classified as biologics by the U.S. FDA, and their regulation is strictly defined. Furthermore, the National Institutes of Health (NIH) has underscored the importance of specialist treatment and monitoring as part of Alpha-1 Antitrypsin Deficiency (AATD) care (Source: National Library of Medicine). This coupled with a reimbursement model favoring hospital purchases makes hospital pharmacies the primary distributors of Alpha-1 Antitrypsin Deficiency (AATD) medications worldwide.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
Regulatory Acceleration of Gene Therapies for AATD (Regulatory + Industry Shift, 2025–2026) |
|
|
High Unmet Need Driving Continued R&D Investment (Macro + Industry Shift, 2024–2026) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
(Source: Tessera Therapeutics, Rare Disease Advisor)
Global Alpha-1 Antitrypsin Deficiency Augmentation Therapy Market Dynamics

To learn more about this report, Request Free Sample
Market Drivers
- Increasing diagnosis rates of genetic disorders through advanced screening programs: The key driver is expanding the identifiable patient population. The World Health Organization suggests that approximately 7.9 million infants, representing roughly 6% of global births annually, are born with genetic or partially genetic disorders. This highlights a significant worldwide health concern (Source: World Health Organization). Moreover, large-scale genomic screening programs have uncovered that nearly 90% of individuals with actionable genetic risks had not been previously diagnosed. This emphasizes the crucial role of screening in uncovering cases that would otherwise go undetected (Source: JAMA Network). The growing adoption of genetic screening is, in turn, significantly improving early diagnosis rates and driving the demand for specialized treatments.
- Growing demand for augmentation therapy as a standard treatment for severe Alpha-1 Antitrypsin Deficiency (AATD) patients to prevent lung function decline: Clinical recommendations from the National Institutes of Health suggest that augmentation therapy can be effective in slowing down the development of emphysema by ensuring adequate levels of alpha-1 antitrypsin in the lungs (Source: National Institutes of Health). Given the rising incidence of severe Alpha-1 Antitrypsin Deficiency (AATD) diagnoses, especially among individuals predisposed to Chronic Obstructive Pulmonary Disease (COPD), physicians are progressively implementing augmentation therapy as a long-term approach to disease management. Consequently, this trend is fueling consistent treatment demand and fostering the market expansion.
Emerging Trends
- Shift Toward Gene and Recombinant Therapies: Gene therapy and recombinant Alpha-1 Antitrypsin (AAT) are becoming more important in research as potential long-lasting or even curative alternatives to treatments derived from plasma. This trend could reduce the need for plasma, thus changing how we approach treatment.
- Expansion of Plasma Collection Infrastructure: The surge in demand for therapies derived from plasma, such as AAT augmentation, is prompting significant investments. These are aimed at establishing new plasma collection centers and sophisticated fractionation facilities, all to secure a reliable and expandable supply chain.
Regional Insights
To learn more about this report, Request Free Sample
Why is North America a Strong Market for Alpha-1 Antitrypsin Deficiency Augmentation Therapy?
North America leads the global Alpha-1 antitrypsin deficiency augmentation therapy market, accounting for an estimated 41.4% share in 2026, owing to its extensive expenditure on healthcare, advanced diagnosis capabilities, and adequate reimbursement facilities. For instance, the U.S. is one of the most advanced countries with regard to healthcare expenditure, with a spending of over USD 14,885 per person on healthcare- the highest healthcare costs per capita across similar countries. Concurrently, Canada spends around USD 7,301 per person on healthcare (Source: Peter G. Peterson Foundation). Moreover, government-sponsored reimbursement programs such as Medicare and Medicaid further ensure better patient affordability.
Why Does Asia Pacific Alpha-1 Antitrypsin Deficiency Augmentation Therapy Market Exhibit High Growth?
The Asia Pacific Alpha-1 antitrypsin deficiency augmentation therapy market is expected to exhibit the fastest growth with an estimated contribution of 20.4% share to the global market in 2026, owing to the improved healthcare infrastructure, increased spending by governments on health care, and better diagnoses of rare disorders. The public spending by countries like China and India on health care is increasing rapidly; for example, health care spending in China is around 5.94% of its GDP, whereas health care spending in India is increasing to reach around 3.34% of the GDP (Source: The World Bank Group). Moreover, scheme like Ayushman Bharat is also contributing in enhancing insurance cover and availability of cutting-edge therapies. Increased knowledge about genetic diseases, greater access to healthcare facilities, and increased adoption of biological therapies like Prolastin-C are also fueling growth in the market.
Global Alpha-1 Antitrypsin Deficiency Augmentation Therapy Market Outlook for Key Countries
Why are the U.S. Leading Innovation and Adoption in the Alpha-1 Antitrypsin Deficiency Augmentation Therapy Market?
The U.S. stands out regarding innovation and adoption of treatments used for alpha-1 Antitrypsin Deficiency (AATD) augmentation therapy owing to its robust regulatory framework, research infrastructure, and access to these therapies. The U.S. FDA encourages innovations related to rare diseases by supporting initiatives such as the orphan drug designation initiative offered under the Orphan Drug Act, where companies receive incentives such as market exclusivity for developing and marketing drugs that help treat conditions such as Alpha-1 Antitrypsin Deficiency (AATD). In addition, the National Institutes of Health encourages research into genetic disorders, including AATD, thereby promoting clinical innovations and new drugs in the pipeline. Consequently, the U.S. is advantaged owing to its already existing market access, in particular, Prolastin-C, owing to reimbursement frameworks such as Medicare and Medicaid.
Is U.K. a Favorable Market for Alpha-1 Antitrypsin Deficiency Augmentation Therapy Market?
The U.K. emerges as a relatively favorable nation for augmentative therapy for AATD due to the robustness of its healthcare system, where all citizens have access to diagnostic tests and rare disease treatments under the umbrella of the National Health Service. Government health expenditures remain considerable, at approximately 11.1% of the Gross Domestic Product in 2024, or an estimated USD 78.8 billion (£58 billion) expenditure in 2023/2024 (Source: Office of National Statistics). These expenditures indicate a substantial commitment to the healthcare sector. Despite this, the adoption of high-cost pharmaceuticals, exemplified by Prolastin-C, is limited, primarily because of the stringent cost-effectiveness evaluations mandated by National Institute for Health and Care Excellence (NICE).
Is Japan Emerging as a Key Growth Hub for the Alpha-1 Antitrypsin Deficiency Augmentation Therapy Market?
The Japan’s economic landscape offers itself as an emerging growth hub for the treatment of Alpha-1 Antitrypsin Deficiency (AATD) via augmentation therapy, given its robust healthcare infrastructure and the financial support schemes provided by the government (e.g., Specified Disease Treatment Research Program, High-Cost Medical Expense Benefit System, Designated Intractable Diseases Subsidy Program). The country spends an estimated amount of between 10.6% and 11.5% of its GDP in the healthcare sector, a figure higher than the 9.3% spent on average by OECD countries. (Source: Organisation for Economic Co-operation and Development)
Additionally, there exists a universal healthcare insurance program that serves up to 100% of the population, thus offering numerous treatment options. Moreover, Japan boasts one of the fastest-growing elderly populations globally, with ~29.3% of its population aged 65 and above (over 36 million people) as of 2024, thereby necessitating biologics for the treatment of any illnesses. (Source: Statistics Bureau of Japan)
Why Does Germany Top the European Alpha-1 Antitrypsin Deficiency Augmentation Therapy Market?
Germany ranks top in the European Alpha-1 antitrypsin deficiency augmentation therapy market owing to its sophisticated healthcare system, efficient diagnoses, and a comprehensive reimbursement policy (e.g., Statutory Health Insurance (SHI), AMNOG benefit assessment framework, sickness fund coverage (Gesetzliche Krankenversicherung - GKV)). The regulatory body in Germany is the Federal Institute for Drugs and Medical Devices, which helps in the quick assessment of these therapies. Furthermore, Germany boasts an extensive network of specialized treatment centers along with the greater awareness of genetically acquired diseases, making it easier to diagnose patients and get started on their therapy.
Is Alpha-1 Antitrypsin Deficiency Augmentation Therapy Market Developing in France?
The Alpha-1 antitrypsin deficiency augmentation therapy market will continue to grow within France owing to the robustness of the healthcare industry there and the availability of effective approaches to manage rare diseases. In France, universal access to specialized medications is provided to everyone by way of the National Health Insurance program; hence, any person affected by a chronic disease or an inheritable condition can easily access treatment for the same. There are several programs and patient registers that make it easy to diagnose and treat the rare disease in question. There are also various systems used to reimburse drugs that include Sécurité Sociale and Affections de Longue Durée among other programs that make it easy to bring down the prices of biological drugs. However, growth of the market is hindered by inefficiencies in diagnosis and regulation of the augmentation therapy.
Regulatory Landscape Governing the Global Alpha-1 Antitrypsin Deficiency Augmentation Therapy Market
|
Region |
Regulatory Authority |
Regulatory Status |
Key Regulation |
Impact on Market |
|
North America |
U.S. Food and Drug Administration |
Well-established and stringent regulatory framework for plasma-derived biologics |
Biologics License Application (BLA), Orphan Drug Act |
Strong regulatory clarity supports product approvals and reimbursement, driving high adoption but increasing compliance costs |
|
Europe |
European Medicines Agency |
Harmonized regulatory environment across EU with focus on rare diseases |
Orphan Medicinal Product Regulation, Good Manufacturing Practices (GMP) |
Encourages innovation through orphan drug incentives while ensuring strict safety and quality standards |
|
Asia Pacific |
National Medical Products Administration, Pharmaceuticals and Medical Devices Agency |
Evolving regulatory frameworks with increasing focus on biologics |
Accelerated approval pathways, biologics guidelines |
Growing regulatory support is improving market access, though variability across countries remains a challenge |
|
Latin America |
Agência Nacional de Vigilância Sanitária |
Developing regulatory systems with gradual alignment to global standards |
Biologics approval pathways, GMP compliance |
Slower approval timelines may limit market penetration, but improving frameworks support future growth |
|
Middle East & Africa |
Saudi Food and Drug Authority, South African Health Products Regulatory Authority |
Emerging regulatory landscape with reliance on international approvals |
Import regulations, biologics guidelines |
Market growth is supported by increasing healthcare investment, though regulatory inconsistencies may hinder rapid adoption |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
How is the development of novel therapies including gene therapy, RNA-based treatments, and recombinant AAT products creating new growth opportunities in the Alpha-1 antitrypsin deficiency augmentation therapy market?
The emergence of innovative therapies like gene therapy, RNA therapy, and recombinant AAT therapies will have a huge impact on the future prospects of the Alpha-1 antitrypsin deficiency augmentation therapy market. As per the U.S. FDA, current treatment options do not completely cater to the requirements of patients, and there is no curative treatment option currently, underscoring the high level of unmet need within the market (Source: Food and Drug Administration).
Emerging approaches such as gene therapy aim to enable long-term or one-time correction of AAT deficiency, while RNA-based therapies and recombinant products offer scalable, non–plasma-dependent alternatives.
In addition, there are various ongoing clinical trials. For instance, the AAV-based gene therapy study for AATD (NCT06996756) listed on ClinicalTrials.gov, showcase increased research efforts aimed at developing more effective and potentially long-term treatment options for patients (Source: ClinicalTrials.gov). These emerging treatment alternatives promise better efficacy and effectiveness, along with increased access, which will result in substantial market growth opportunities.
Market Players, Key Development, and Competitive Intelligence

To learn more about this report, Request Free Sample
Key Developments
- In December 2025, Regeneron Pharmaceuticals, Inc. and Tessera Therapeutics unveiled a collaboration. The goal is developed TSRA-196, a gene editing therapy still in the investigational stage, targeting Alpha-1 Antitrypsin Deficiency (AATD). This partnership underscores the increasing attention the industry is giving to gene-based therapies, specifically those designed to tackle the genetic root of AATD.
- In October 2025, Sanofi revealed that its experimental treatment, efdoralprin alfa, had successfully achieved all primary and key secondary objectives in a Phase 2 trial targeting emphysema linked to alpha-1 antitrypsin deficiency (AATD). The therapy showed greater improvements in functional AAT levels than the standard plasma-derived augmentation therapy, and it also requires less frequent administration. The observed advancements highlight the growing prevalence of recombinant therapies.
Competitive Landscape
The Alpha-1 antitrypsin deficiency augmentation therapy market is highly concentrated, with competition dominated by a few key players such as Grifols, CSL Behring, and Takeda Pharmaceutical Company due to high entry barriers linked to plasma sourcing and complex manufacturing. The competitiveness of this market sector lies in robust collection systems, reliability of products, and proven efficacy of the same in clinical use. Increasingly, companies are beginning to concentrate on enlarging fractionation facilities along with ensuring the consistency of supplies to cater to increasing demand. Recombinant protein technology research is likely to have an impact in future. Key focus areas include:
- Expansion of plasma collection and fractionation capacity
- Development of recombinant and gene-based alternatives
- Strengthening global distribution and cold chain logistics
Market Report Scope
Alpha-1 Antitrypsin Deficiency Augmentation Therapy Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1,878.2 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 3.94% | 2033 Value Projection: | USD 2,461.9 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Grifols S.A., CSL Behring, Takeda Pharmaceutical Company Limited, Kamada Ltd., Baxter International Inc., LFB Biotechnologies, Pfizer Inc., AstraZeneca plc, Arrowhead Pharmaceuticals, Inc., and Vertex Pharmaceuticals Incorporated |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Analyst Opinion (Expert Opinion)
- The trajectory of the Alpha-1 antitrypsin deficiency augmentation therapy market seems promising in indications related to pulmonary conditions (emphysema/COPD) in the U.S., where diagnostic rate, reimbursement and usage are much greater. Europe will remain a significant market because of the rare disease programs that exist, with emerging potential in countries like UK, and Germany because of improved diagnostic rates, but penetration is lower compared to Europe.
- The most significant potential resides in intravenous (IV) augmentation therapy for pulmonary conditions, specifically chronic obstructive pulmonary disease (COPD) and emphysema, within the United States. This is primarily due to elevated diagnostic rates, robust reimbursement structures, and the extensive utilization of plasma-derived AAT therapies. Moreover, the ongoing need in this area is supported by clinical guidelines from organizations like the American Thoracic Society and the Global Initiative for Chronic Obstructive Lung Disease.
- For the market players to gain a competitive advantage, they must concentrate on building up their plasma collection network for supply assurance, exploring advanced treatments like gene therapy and recombinant products, and improving their patient identification campaigns through screening programs. Collaboration with healthcare organizations and rare disease groups, as well as geographical diversification by tapping into regions where rare diseases remain underdiagnosed, would also be crucial.
Market Segmentation
- Drug Insights (Revenue, USD Mn, 2021 - 2033)
- Glassia
- Aralast NP
- Prolastin C
- Trypsone
- Zemaira
- Late Phase Drug
- Route of Administration Insights (Revenue, USD Mn, 2021 - 2033)
- Inhalational
- Injections
- Distribution Channel Insights (Revenue, USD Mn, 2021 - 2033)
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
- Regional Insights (Revenue, USD Mn, 2021 - 2033)
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
- Key Players Insights
- Grifols S.A.
- CSL Behring
- Takeda Pharmaceutical Company Limited
- Kamada Ltd.
- Baxter International Inc.
- LFB Biotechnologies
- Pfizer Inc.
- AstraZeneca plc
- Arrowhead Pharmaceuticals, Inc.
- Vertex Pharmaceuticals Incorporated
Sources
Primary Research Interviews
- Alpha-1 Antitrypsin Deficiency augmentation therapy manufacturers and plasma-derived biologics providers
- Pulmonologists and respiratory specialists treating COPD and emphysema linked to AATD
- Hospital pharmacists and infusion therapy providers administering IV augmentation therapy
- Clinical researchers and rare disease experts specializing in genetic and respiratory disorders
Stakeholders
- Providers of Alpha-1 Antitrypsin Deficiency augmentation therapies
- End-use Sectors
- Hospitals and specialty clinics (Pulmonology)
- Infusion centers
- Research institutes
- Pharmaceutical & biotechnology companies
- Regulatory & Health Bodies: World Health Organization, U.S. Food and Drug Administration, European Medicines Agency, National Institutes of Health
Databases
- FDA – Approved biologics and blood products database (Alpha-1 Proteinase Inhibitors approvals)
- WHO – Global Health Observatory (GHO)
- NIH – Rare disease and clinical trials database
- OECD Health Statistics – Healthcare expenditure and rare disease burden
Magazines
- Pharmaceutical Technology – Plasma-derived therapies and biologics manufacturing trends
- BioPharma Dive – Rare disease therapeutics and biologics pipeline developments
- Genetic Engineering & Biotechnology News – Advances in protein therapeutics and gene therapy alternatives
- Healthcare Business Today – Rare disease market access and reimbursement trends
Journals
- Journal of Translational Medicine – Rare disease and biologics therapy research
- The Lancet Respiratory Medicine – Clinical management of COPD and AATD
- American Journal of Respiratory and Critical Care Medicine – AATD treatment studies
- Orphanet Journal of Rare Diseases – Epidemiology and treatment of AATD
Newspapers
- The Wall Street Journal – Biopharmaceutical investments in rare diseases
- Financial Times – Global biologics and plasma therapy market trends
- The Economic Times – India rare disease and biologics ecosystem
- Business Standard – Biopharma manufacturing and plasma therapy developments
Associations
- Alpha-1 Foundation
- American Thoracic Society (ATS)
- European Respiratory Society (ERS)
- International Federation of Pharmaceutical Manufacturers & Associations (IFPMA)
Public Domain Sources
- WHO – Guidelines on genetic and rare diseases
- NIH – Alpha-1 Antitrypsin Deficiency research initiatives
- FDA – Approved augmentation therapies such as GLASSIA and ZEMAIRA for AATD treatment
- EMA – Orphan drug designation and biologics regulation
Proprietary Elements
- CMI Data Analytics Tool, Proprietary CMI Existing Repository of information for last 10 years.
Share
Share
About Author
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients