Europe Cystoscopy Market Size and Forecast – 2026 to 2033
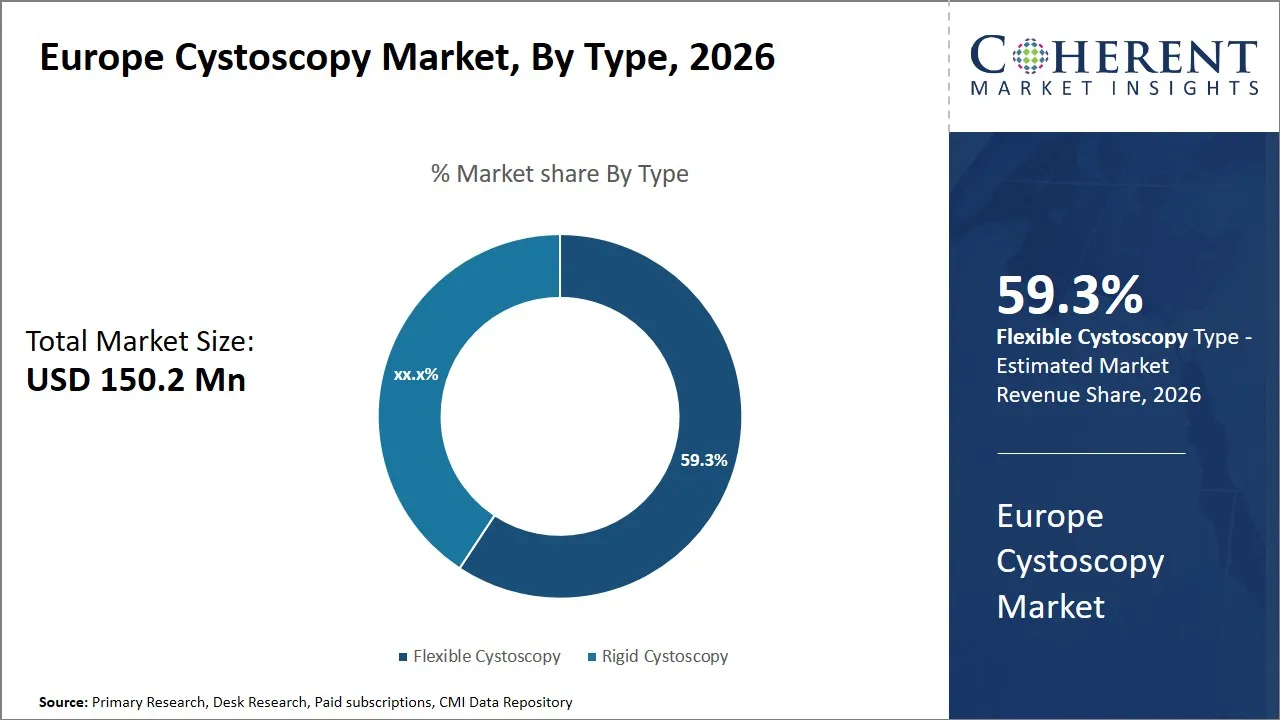
The Europe Cystoscopy Market is estimated to be valued at USD 150.2 Mn in 2026 and is expected to reach USD 199.9 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 5.8% from 2026 to 2033. This steady growth is driven by the increasing prevalence of urological disorders, technological advancements in cystoscopic devices, and rising awareness among healthcare professionals regarding early diagnosis and treatment of bladder-related conditions.
Key Takeaways of the Europe Cystoscopy Market
- Flexible cystoscopy segment is expected to lead the Europe cystoscopy market, capturing 59.3% share in 2026.
- Hospitals segment is estimated to represent 38.1% of the Europe cystoscopy market share in 2026.
Market Overview
- Cystoscopy remains a core procedure in European clinical practice for non-muscle-invasive bladder cancer follow-up, which keeps baseline procedural demand structurally strong across hospitals and urology centers.
- Demand is gradually moving from basic white-light systems toward HD, 4K, and advanced visualization platforms, especially where clinicians want better lesion detection, documentation, and procedural efficiency in bladder cancer management.
- The core demand engine is bladder cancer surveillance, where cystoscopy remains central to follow-up in European clinical practice. That gives the market recurring procedure volume and makes it more resilient than device categories tied only to first-time diagnosis.
- The market is being pushed by HD imaging, better visualization, and broader-use cystoscopy platforms, including recent CE-mark expansion for single-use flexible cysto-nephroscopy solutions in Europe.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
Rising bladder cancer surveillance demand across Europe |
|
|
Shift toward outpatient and office-based cystoscopy procedures |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Why Does the Flexible Cystoscopy Segment Dominate the Europe Cystoscopy Market in 2026?
The flexible cystoscopy segment is expected to hold the largest share of 59.3% in 2026, primarily attributed to technological advancements that emphasize patient comfort and procedural efficiency. Flexible cystoscopes are minimally invasive in nature, and they can be maneuvered through the urinary tract easily as compared to their rigid counterparts. Such flexibility greatly minimizes the discomfort and pain caused during examinations and hence the most preferred when it comes to patients and clinicians.
Moreover, flexibility of the cystoscope allows the achievement of greater diagnostic accuracy and treatment. Physicians are able to routinely check and perform focused procedures on the bladder and urethra using their capability to handle the intricate anatomical curves of these organs with minimal chances of causing mucosal damage. This is particularly important to patients who need serial cystoscopic assessment (i.e., patients with frequent urinary tract infections or monitoring bladder cancer, etc.) and who would benefit by having less traumatizing method.
For instance, in October 2023, Ambu launched the aScope 5 Cysto HD in Europe, introducing a single-use HD flexible cystoscopy solution aimed at improving workflow efficiency and reducing reprocessing burden in urology settings.
Why Does the Hospitals Segment Dominate the Europe Cystoscopy Market in 2026?
The hospitals segment is projected to capture 38.1% share in 2026, due to their pivotal role in providing comprehensive urological care. Hospitals equipped with modern infrastructure and teams with high level of competencies are well-prepared to conduct a comprehensive range of cystoscopic procedures, both of regular diagnostics and more complicated therapeutic programs. This is what renders hospitals the choice of location of cystoscopy procedures.
The main factor contributing to hospital’s dominance is the fact that in the recent times there has been a growing trend of urological diseases that have to undergo cystoscopic investigation, such as bladder cancer, urinary tract infections and benign prostatic hyperplasia. Such cases are usually referred to hospitals where more complex and high-risk patients are treated and where they can get a lot of diagnostic workup and follow-up. Their ability to provide one stop solution care, such as imaging, biopsy, and surgery, is an incentive to the patients and referring physicians to use hospital-based cystoscopy services.
Advancements in Next-Generation Cystoscopy Technologies
- The next-gen cystoscopy development in Europe is heading in two distinct directions; towards improved visualization and simplified provision of care. Vendors are also going beyond conventional white light to HD and blue light enabled platform on the visualization front, and flexible cystoscopy is also gaining a higher priority as much of the bladder cancer monitoring occurs in the outpatient environment. A good example is the Photocure-Richard Wolf deal of coming with a high-definition flexible blue light cystoscope to be used in screening, diagnosis, and surveillance procedures, particularly in outpatients.
- Meanwhile, workflow and the economics of infection control are influencing product development. Single-use cystoscopy systems are being established as an effective method of reducing the re-processing load and providing a regular scope performance that is most valuable in office-based care and infection-prone settings. Meanwhile, AI is beginning to transition out of research and to early clinical discussion, at the European conferences systems are demonstrated that can identify bladder lesions that are difficult to see on cystoscopic images, and this is where capabilities like AI are going to be within the next product generations.
Market Players, Key Developments, and Competitive Intelligence
To learn more about this report, Request Free Sample
Key Developments
- In December 2025, Olympus Europa announced the next evolution in rigid cystoscopes, marking a Europe-focused product development in the cystoscopy market. The announcement indicates continued investment in premium reusable cystoscopy systems across the region, even as demand also grows for flexible and single-use formats.
- In May 2025, Ambu reported that it strengthened its urology portfolio through a CE mark expansion for Ambu aScope 5 Cysto HD, enabling use for cysto-nephroscopy procedures. For the Europe cystoscopy market, this is important because it broadens the addressable procedure base of a single-use HD cystoscopy platform and reinforces the competitive push toward more versatile disposable endoscopy offerings.
- In March 2025, Photocure announced that two abstracts presented at the European Association of Urology 2025 Congress highlighted the clinical value of Blue Light Cystoscopy in bladder cancer, including recurrence-risk implications and support for better management decisions. This matters for Europe because EAU is one of the region’s most influential urology platforms, and positive congress visibility supports wider hospital adoption of advanced cystoscopy techniques across European centers.
Top Strategies Followed by Europe Cystoscopy Market Players
|
Player Type |
Strategic Focus |
Examples |
|
Established Market Leaders |
Leading companies compete through continuous R&D, focusing on better visualization, improved patient comfort, and minimally invasive performance in cystoscopy systems. They also strengthen their market position through strategic partnerships, OEM collaborations, and wider service networks, which help speed up product development, improve market reach, and support long-term growth across Europe and other regions. |
Photocure and Richard Wolf partnered to develop a next-generation high-definition flexible blue light cystoscopy solution, and in February 2025 they introduced an interim flexible solution in cleared European markets. This is a clear example of how collaboration helps companies accelerate innovation and expand advanced cystoscopy adoption in Europe. |
|
Mid-Level Players |
Mid-level players in the Europe cystoscopy market usually compete on value rather than premium positioning. They focus on reliable, cost-effective systems that meet core clinical needs, while using OEM partnerships, component collaboration, and incremental product improvements to strengthen quality, expand reach, and stay relevant in price-sensitive segments such as public hospitals and outpatient clinics. |
Vimex Sp. z o.o. is a good example of this strategy. The company presents itself as an original manufacturer of rigid endoscopy devices, offers urology solutions, and supports OEM and R&D cooperation for partners. That shows how a mid-tier player can combine affordable endoscopy products with manufacturing collaboration to improve market presence and build brand credibility. |
|
Small-Scale Players |
Small-scale players in the Europe cystoscopy market usually compete by focusing on niche innovation rather than scale. They target specific needs such as single-use cystoscopes, portable imaging systems, or simplified digital workflows, and often rely on local partnerships or regional clinical collaborations to improve visibility, adapt to market needs, and build presence in selected countries or healthcare centers. |
UroViu is a strong example of this approach. The company focuses on single-use cystoscopy and promotes portable, office-friendly platforms such as the Uro-GHD and Uro-V, which are designed around workflow simplicity, consistent scope quality, and reduced reprocessing burden. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Impact of National Healthcare Reimbursement Rates on the Europe Cystoscopy Market
- One of the largest demand levers that are hidden in the Europe cystoscopy market is the rate of national healthcare reimbursement since it dictates the location of cystoscopy and what hospitals can afford to justify their purchase. Europe Reimbursement is not a single system by country, and the activity of the hospital is usually reimbursed using DRG-style systems and outpatient services are reimbursed using individual fee schedules or tariffs. Among other things, England sets its prices under the NHS Payment Scheme using national tariff structure, HRG-linked pricing structures or best-practice tariff structure that can affect provider behavior and site of care.
- This fragmentation has a direct influence on procurement and technology adoption. Under tight outpatient tariffs, providers are inclined to high-throughput workload, fleets with high durability and low cost, and to contractual agreements that are price-controlled; in contrast, where payment systems are incentivized towards high quality or increased complexity work, hospitals are more eager to adopt more advanced visualization and new systems. The same dynamic is reflected in other sizeable European markets: outpatient reimbursement in Germany is based on the EBM catalog, which is updated periodically, and outpatient reimbursement in France is based on the CCAM procedural nomenclature, which is linked to commercial attractiveness of particular procedures and technologies, and can therefore change them quickly.
Market Report Scope
Europe Cystoscopy Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 150.2 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.8% | 2033 Value Projection: | USD 199.9 Mn |
| Segments covered: |
|
||
| Companies covered: |
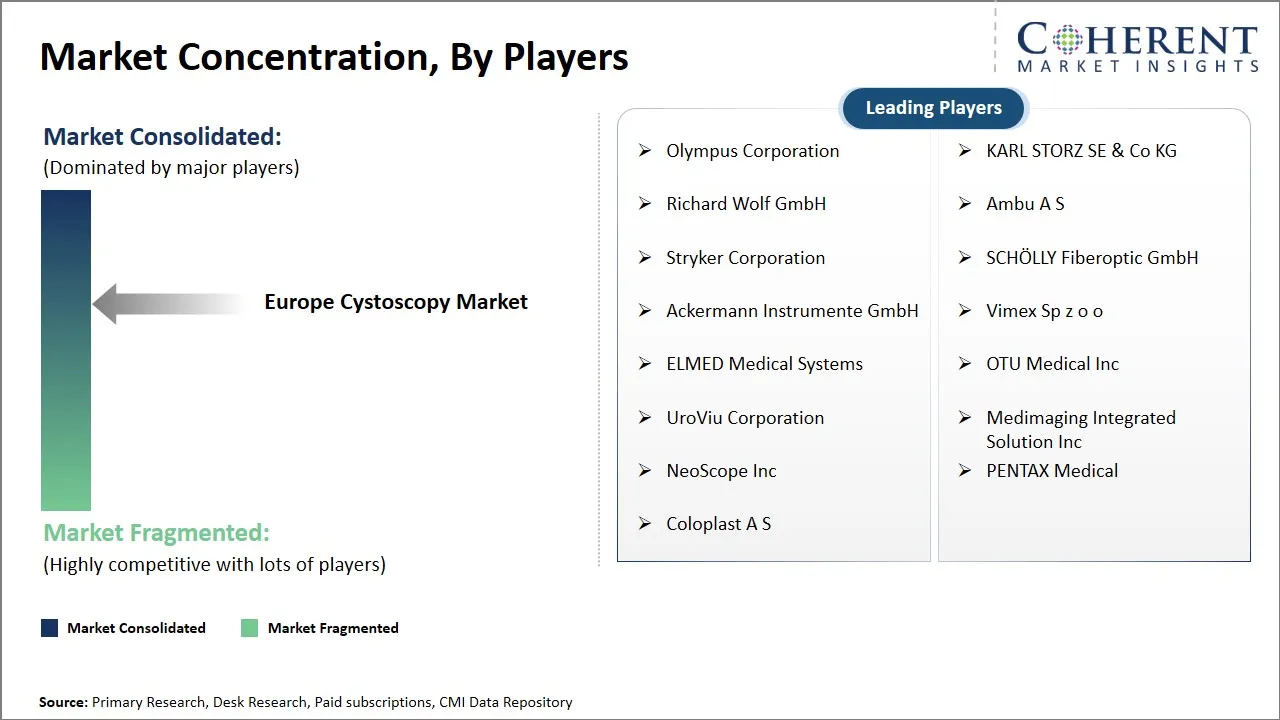
Olympus Corporation, KARL STORZ SE & Co KG, Richard Wolf GmbH, Ambu A S, Stryker Corporation, SCHÖLLY Fiberoptic GmbH, Ackermann Instrumente GmbH, Vimex Sp z o o, ELMED Medical Systems, OTU Medical Inc, UroViu Corporation, Medimaging Integrated Solution Inc, NeoScope Inc, PENTAX Medical, and Coloplast A S |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Europe Cystoscopy Market Dynamics
To learn more about this report, Request Free Sample
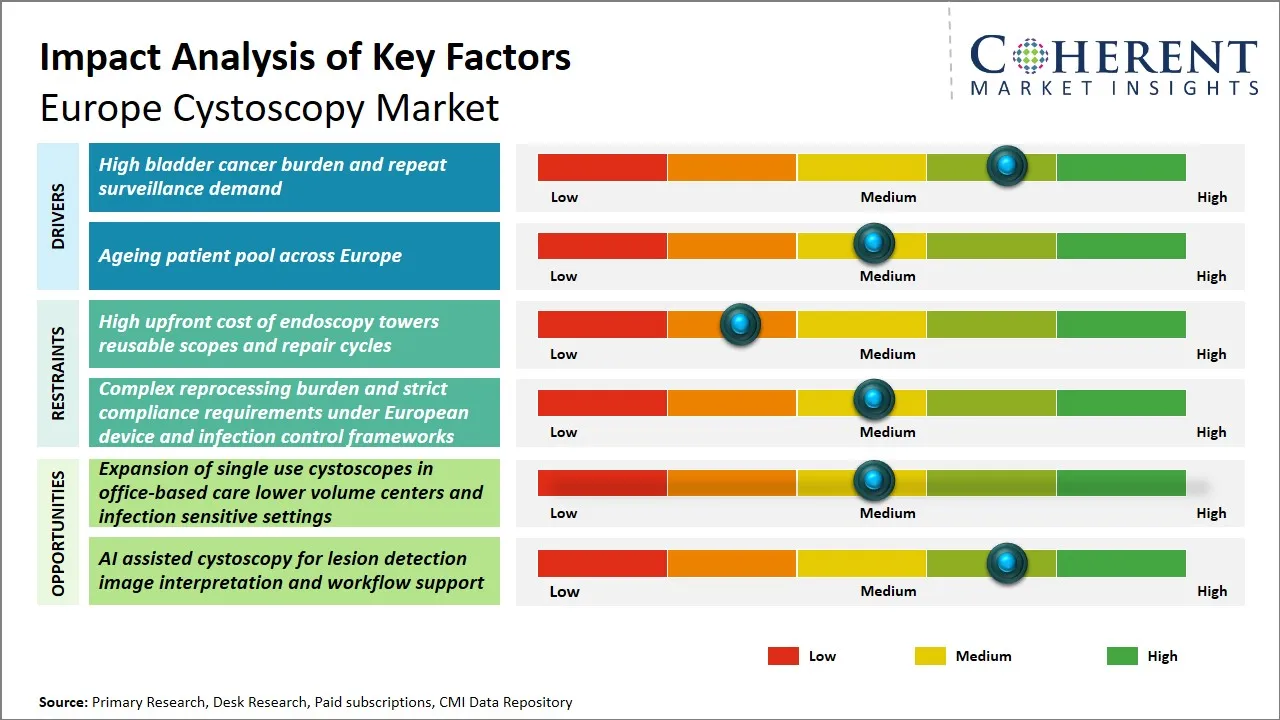
Europe Cystoscopy Market Driver - High Bladder Cancer Burden and Repeat Surveillance Demand
The increase of bladder cancer in the different parts of Europe is a major factor that is fueling the need to undertake cystoscopy tests because the given illness requires continuous observation and diagnostic management. Since bladder cancer is a disease with high recurrence rates, patients need periodic follow-ups to identify the relapse or further development of the tumor at its earliest stage.
This occurrence rate is quite high and cystoscopy is thus an inseparable part of the clinical management pathway and creates a sustained demand on cystoscopic equipment and related technologies. Moreover, the sensitivity of cystoscopy has been increased by the development of improved imaging and flexible scope which is more comfortable to the patient during the surveillance process and thus has led to the adoption of such technology.
For instance, in April 2023, UroViu launched the Uro-GHD, a high-definition sterile single-use cystoscope designed to improve visualization, patient comfort, and office-based workflow efficiency.
Europe Cystoscopy Market Opportunity - Expansion of Single-Use Cystoscopes in Office-Based Care and Infection-Sensitive Settings
Europe cystoscopy market is also experiencing massive opportunity owing to the growing trend towards the adoption of single-use cystoscopes especially in office-based care and low volume healthcare facilities. The conventional reusable cystoscopes may need extensive sterilization procedures that are time consuming, expensive and prone to cross contamination and transmission of the infection. The idea of single-use cystoscopes is quite promising, as it gives sterile, ready-to-use cystoscopes, which do not require reprocessing and, due to this feature, improve patient safety in the context of the environment with the high risk of infection exposure, such as outpatient clinics and small healthcare facilities. Moreover, the increased focus on infection control following the strict healthcare regulations and the increased awareness of healthcare-associated infections (HAIs) is driving the disposable devices demand.
For instance, in December 2025, Asieris announced its CE application submission for APLD-2304, a portable single-use blue-light flexible cystoscope. This is a direct instance of single-use cystoscopes expanding toward office-based and infection-sensitive use cases by combining portability with sterile, one-time-use workflow to reduce reprocessing dependence.
Analyst Opinion (Expert Opinion)
- Europe cystoscopy market has been expanding based on the stable demand in bladder cancer surveillance, ageing patient population and the upgrades ongoing towards HD, 4K, blue light and single use cystoscopy systems. Outpatient and office-based care is the actual opportunity where providers seek faster workflow, reduced reprocessing load, and stable scope. The only problem is that Europe is a very regulated and price-conscious market, and manufacturers must have good clinical evidence, MDR compliance and apparent value offer to succeed.
- Conferences like the EAU Congress 2024 and 2025 and EMUC 2024 have been used in the recent times to move the discussion on bladder cancer surveillance, imaging innovation, and practical implementation of more advanced cystoscopy platforms in Europe. It is also evidenced by recent developments such as Photocur’s blue light cystoscopies development, CE-mark expansion by Ambu with aScope 5 Cysto HD, and CE certification of UroViu, which indicate that the market is shifting towards more sophisticated and outpatient-friendly and single-use solutions.
Market Segmentation
- Type Insights (Revenue, USD Mn, 2021 - 2033)
- Flexible Cystoscopy
- Rigid Cystoscopy
- End User Insights (Revenue, USD Mn, 2021 - 2033)
- Hospitals
- Ambulatory Surgical Centers
- Specialty Clinics
- Cancer Centers/ Diagnostic Centers
- Others
- Key Players Insights
- Olympus Corporation
- KARL STORZ SE & Co KG
- Richard Wolf GmbH
- Ambu A S
- Stryker Corporation
- SCHOLLY Fiberoptic GmbH
- Ackermann Instrumente GmbH
- Vimex Sp z o o
- ELMED Medical Systems
- OTU Medical Inc
- UroViu Corporation
- Medimaging Integrated Solution Inc
- NeoScope Inc
- PENTAX Medical
- Coloplast A S
Sources
Primary Research Interviews
- Industry Stakeholders list
- Consultant Urologists
- Uro-oncology Specialists
- Hospital Procurement Heads
- Endoscopy Unit Managers
- Medical Device Regulatory Affairs Specialists
- Hospital Sterile Services and Reprocessing Leads
End-users list
- Public Hospital Urology Departments
- Private Urology Clinics
- Ambulatory Surgical Centers
- Day-care Endoscopy Units
- Urology Nurses
- Bladder Cancer Patient Advocacy Representatives
Government and International Databases
- Eurostat Health Database
- Eurostat Population and Demography Database
- WHO Regional Office for Europe
- IARC Global Cancer Observatory GLOBOCAN Europe Fact Sheet
- OECD Health at a Glance Europe
- OECD EU Country Health Profiles Dashboard
Trade Publications
- Urology Times
- MedTech Dive
- Medical Design & Outsourcing
- Medscape Urology
- pharmaphorum
- Medical Device Network
Academic Journals
- European Urology
- European Urology Oncology
- European Urology Open Science
- BJU International
- World Journal of Urology
- Journal of Clinical Urology
Reputable Newspapers
- Financial Times
- The Guardian
- The Times
- Le Monde
Industry Associations
- European Association of Urology
- European Association of Urology Nurses
- EAU Section of Oncological Urology
- British Association of Urological Surgeons
- European Society of Urogenital Radiology
- MedTech Europe
Public Domain Resources
- ClinicalTrials.gov
- EU Clinical Trials Information System public portal
- NICE Guidance
- OpenAlex
Proprietary Elements
- CMI Data Analytics Tool: Proprietary analytics tool to analyze real-time market trends, consumer behavior, and technology adoption in market
- Proprietary CMI Existing Repository of Information for Last 8 Years
Share
Share
About Author
Manisha Vibhute is a consultant with over 5 years of experience in market research and consulting. With a strong understanding of market dynamics, Manisha assists clients in developing effective market access strategies. She helps medical device companies navigate pricing, reimbursement, and regulatory pathways to ensure successful product launches.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients