The Bulimia Nervosa Treatment Market is anticipated to grow at a CAGR of 5.2% with a market value of approximately USD 644.8 Mn in 2026 and is expected to reach around USD 964.6 Mn in 2033. The growth is driven by rising prevalence of eating disorders, increasing mental health awareness, improved diagnosis rates, expansion of behavioral therapy and pharmacological treatment options, and growing healthcare access and support infrastructure for psychiatric conditions globally.
Increasing preference for pharmacotherapy and psychotherapy that are backed by scientific research is one of the key factors propelling the market for bulimia nervosa treatments, specifically the usage of SSRI (like fluoxetine) as the primary medication.
Advances in drug treatments for depression, particularly SSRI medication, have proven highly effective, evidenced by research that indicates up to a 50% to 60% reduction in binge-purging behaviors in patients taking fluoxetine versus those on placebo treatment.
Growing awareness and improved diagnosis diagnoses of eating disorders are rapidly increasing the market size for treatment solutions, especially in North America and Europe. As per the WHO, about 9% of the world’s population suffers from eating disorders during their lifetime, indicating an immense potential for untreated patients who can benefit from either drug-based or behavior-based treatment.
Furthermore, increased screening initiatives and mental health awareness campaigns have led to earlier diagnosis, improving treatment initiation rates and supporting long-term pharmaceutical and therapy-based revenue growth.
|
Current Event |
Description and its Impact |
|
Rising U.S. Mental Health Coverage Expansion and Insurance Parity Enforcement (2025–2026) |
|
|
Growing FDA Approvals and Generic Drug Expansion in SSRIs (2025–2026) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Drug Treatment is forecasted to hold the highest market share among all bulimia nervosa treatments in 2026, estimated at around 70%. The predominance of drug treatments in the bulimia nervosa treatment market is attributed to the high clinical dependence on antidepressants, primarily selective serotonin reuptake inhibitors like fluoxetine.
Furthermore, studies conducted under clinical trials within the National Library of Medicine (PubMed Central) show that fluoxetine is able to lower binge–purge behaviors by 50%–60% relative to placebo, proving its powerful clinical effects and broad use in the medical field.
In March 2024, the National Institute for Health and Care Excellence (NICE) updated clinical guidance reinforcing the use of Selective Serotonin Reuptake Inhibitors (SSRIs), particularly fluoxetine, as a key pharmacological option for bulimia nervosa management in adults.
To learn more about this report, Request Free Sample
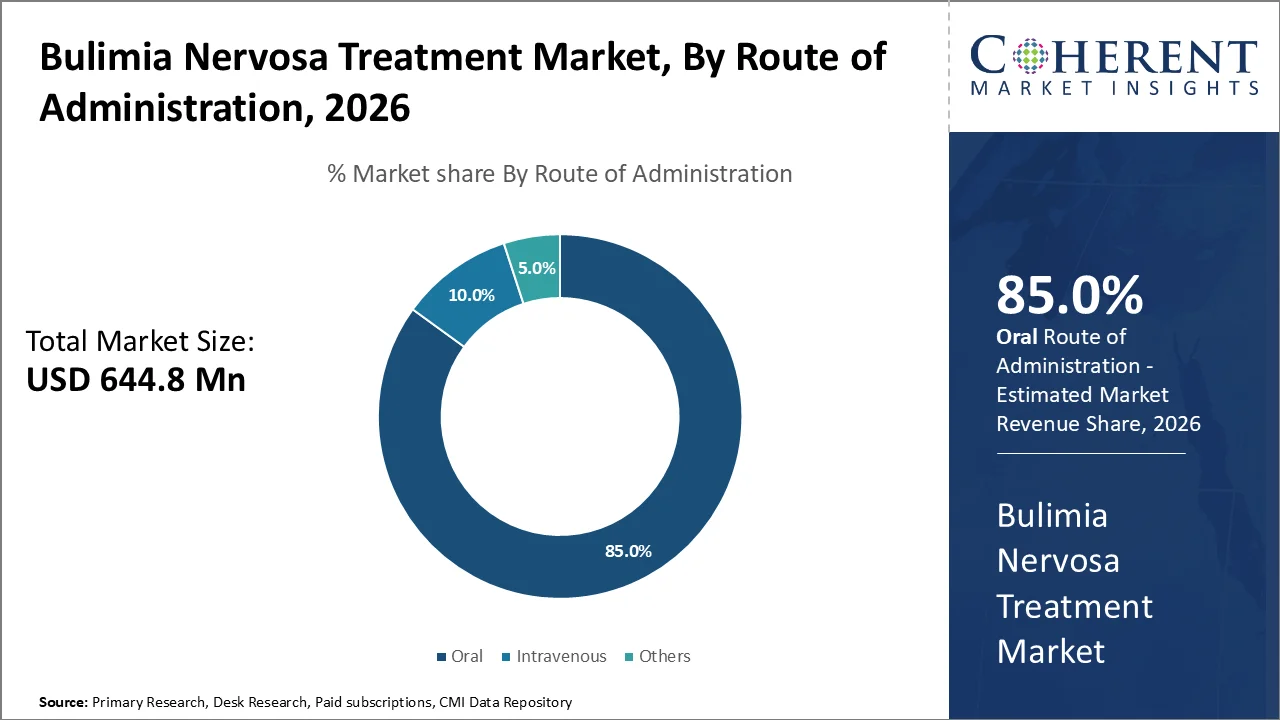
Oral medication forms will lead to maximum revenue shares, accounting for an estimated 85% of the overall administration route segment by 2026. This is due to high utilization of oral medicines including SSRI, SNRI, and tricyclic antidepressant classes that are considered as the main drugs used for treating bulimia nervosa disorders.
This is highly emphasized by the World Health Organization (WHO), pointing out that disorders in mental health are mostly handled through outpatient management programs, and oral medications stand out as the most convenient mode of treatment because of their accessibility and cost-effectiveness among other reasons.
The retail pharmacies distribution channel is expected to occupy the maximum market share in the bulimia nervosa treatment market in 2026, contributing about 50% to the total distribution channel category. The reason behind such high contribution lies in the high repeat filling rate of prescriptions along with the chronicity of the disease which demands lifelong pharmacotherapy with antidepressants including SSRIs.
In May 2025, CVS Health announced further network restructuring and store optimization, including the closure of select underperforming pharmacy locations while expanding pharmacy service models in high-demand areas.
To learn more about this report, Request Free Sample
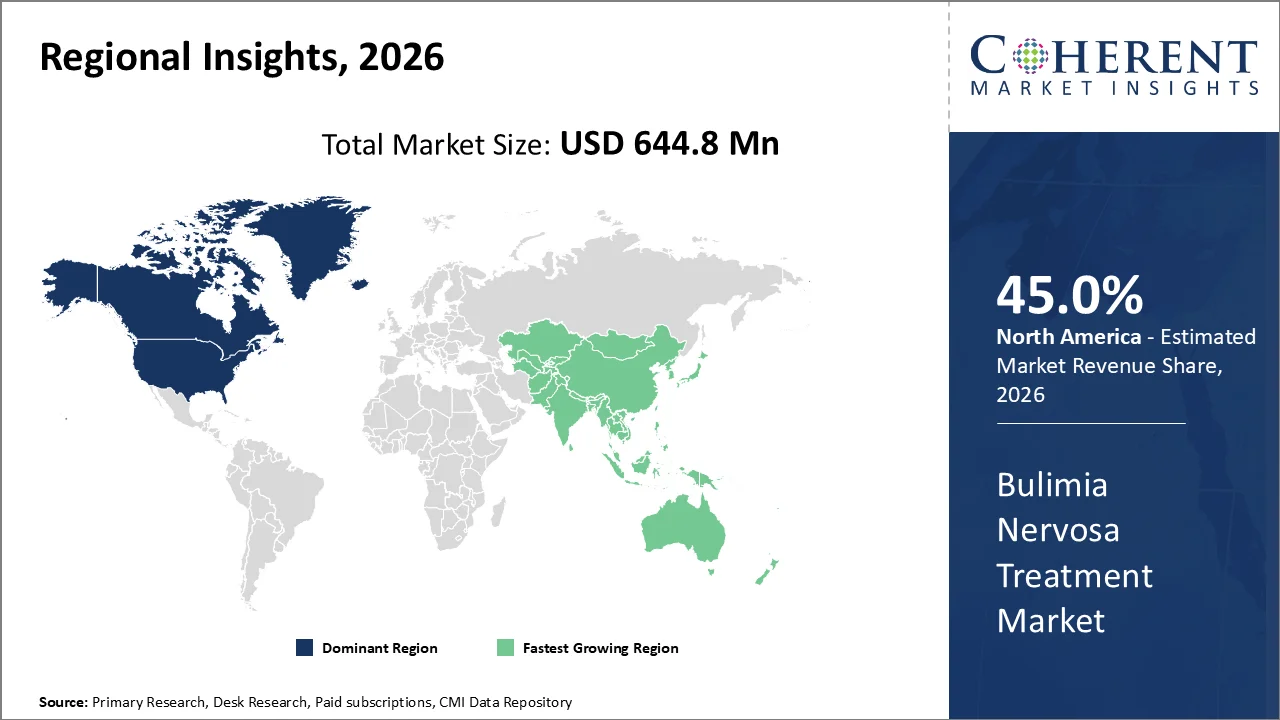
North America is anticipated to hold 45% market share by 2026 due to high awareness levels regarding eating disorders, developed psychiatric care facilities, and improved diagnosis rates. North America is well-developed in terms of behavioral healthcare facilities, and its insurance coverage for all types of mental disorders allows for better access to both medication-based therapy and psychotherapy.
Digital mental health expansion is also accelerating growth, with the U.S. Department of Health & Human Services reporting a more than 150% rise in telehealth-based mental health consultations post-pandemic, improving access to CBT and psychiatric care across urban and rural populations.
In October 2025, CVS Health released its annual pharmacy outlook report highlighting continued dominance of retail pharmacies in the U.S. prescription ecosystem, with strong growth in chronic disease medication dispensing, including mental health drugs such as antidepressants.
Asia-Pacific is estimated to be the fastest growing market during the forecast period of 2026–2033 owing to higher prevalence of mental disorder conditions, growing infrastructure for healthcare services, and growing cases of eating disorders among developing countries including China, India, Japan, and South Korea. This region is experiencing a fast transformation from under-diagnosis to treatment acceptance.
Based on statistics, India alone reports more than 1.5 million cases of eating disorders, whereas Japan and China are witnessing an increase in prevalence rates because of shifts in lifestyle and psychological stress among the youth population.
In September 2025, the University of Oxford introduced “Digital CBTe,” a structured digital cognitive behavioral therapy program for eating disorders, including bulimia nervosa, aimed at improving treatment accessibility in Asia-Pacific and other underserved regions.
The U.S. bulimia nervosa treatment market is projected to continue being the biggest and most developed market over the forecast period. The growth of this market will be attributed mainly to awareness, well-developed facilities for psychiatric care, prevalence of the diagnosis, and wide availability of insurance coverages.
In March 2025, the U.S. Food and Drug Administration (FDA) reaffirmed fluoxetine (Prozac) as the only approved medication specifically indicated for bulimi nervosa, maintaining its status as the standard pharmacological therapy in the United States.
China will become one of the fastest-growing markets in the Asia-Pacific region, attributed to increased mental wellness awareness, elevated levels of stress in cities, improved healthcare facilities, and the growing adoption of telehealth practices.
In September 2025, Xi’an Jiaotong-Liverpool University (China collaboration forum) highlighted that eating disorder prevalence in China is rising rapidly, with a strong increase among adolescents and young adults, particularly in urban regions.
Some of the major key players in Bulimia Nervosa Treatment are Allergan, Inc., Eli Lilly and Company, AstraZeneca plc, Alembic Pharmaceuticals Ltd, Bristol-Myers Squibb Company, GlaxoSmithKline plc, Pfizer Inc., Johnson & Johnson Services, Inc., Dr. Reddy’s Laboratories Limited, Lupin Pharmaceuticals, Inc., Aurobindo Pharma, Zydus Cadila, Apotex Inc., Teva Pharmaceutical Industries Ltd, and Sun Pharmaceutical Industries Ltd.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 644.8 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.2% | 2033 Value Projection: | USD 964.6 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Allergan, Inc., Eli Lilly and Company, AstraZeneca plc, Alembic Pharmaceuticals Ltd, Bristol-Myers Squibb Company, GlaxoSmithKline plc, Pfizer Inc., Johnson & Johnson Services, Inc., Dr. Reddy’s Laboratories Limited, Lupin Pharmaceuticals, Inc., Aurobindo Pharma, Zydus Cadila, Apotex Inc., Teva Pharmaceutical Industries Ltd, and Sun Pharmaceutical Industries Ltd |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Interviews with psychiatrists, clinical psychologists, psychiatric nurses, eating disorder specialists, hospital pharmacy managers, and mental health researchers involved in bulimia nervosa treatment and care delivery systems
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients