Cancer Registry Software Market Size and Forecast – 2026 – 2033
The Global Cancer Registry Software Market size is estimated to be valued at USD 0.24 billion in 2026 and is expected to reach USD 0.46 billion by 2033, exhibiting a compound annual growth rate (CAGR) of 9.6% from 2026 to 2033.
Global Cancer Registry Software Market Overview
Cancer registry software is a specialized digital system used to collect, manage, and analyze data on cancer cases. It helps hospitals, cancer centers, and public health agencies systematically record patient information, diagnosis details, tumor characteristics, treatment plans, and outcomes. By centralizing this information, the software ensures accurate data tracking, improves reporting efficiency, and supports compliance with national and regional cancer registry standards. It also enhances data quality through automated validation tools, standardized coding systems, and real-time error checks.
Beyond data collection, cancer registry software plays a crucial role in cancer surveillance and research. It enables healthcare providers and policymakers to identify disease patterns, monitor treatment effectiveness, and track survival rates. The insights generated from these registries support evidence-based decision-making, resource planning, and the development of targeted cancer control programs. Additionally, advanced systems offer analytics dashboards, interoperability with electronic health records, secure cloud-based storage, and customizable reporting features to strengthen public health outcomes.
Key Takeaways
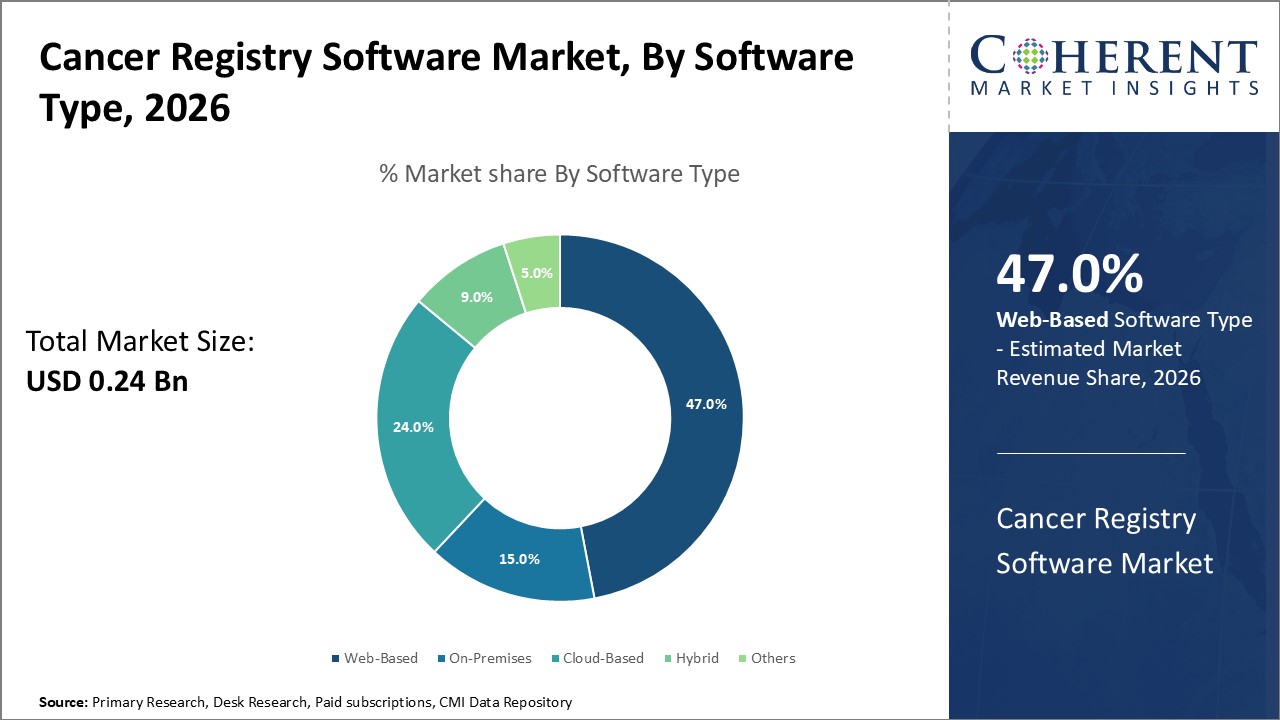
In the software type segment, Web-based solutions lead with 47% share due to scalability and easy hospital integration. Cloud-based platforms grow fastest for flexible, cost-efficient access. On-premises usage declines, while hybrid and other customized deployments serve organizations with mixed or specialized needs.
In the end user segment, Hospitals lead due to large patient data volumes and strict compliance needs. Research institutions grow fastest with rising oncology funding and multi-center studies. Government agencies use registries for surveillance, while pharma and biotech support clinical development. NGOs and smaller providers form the remaining user base.
Within functionality, Data Collection & Management dominates as the core for accurate, standardized cancer data. Analytics & Visualization grows fastest with AI-enabled insights. Reporting & Compliance supports regulatory needs, Patient Tracking aids survivorship, and other specialized modules address additional workflow requirements.
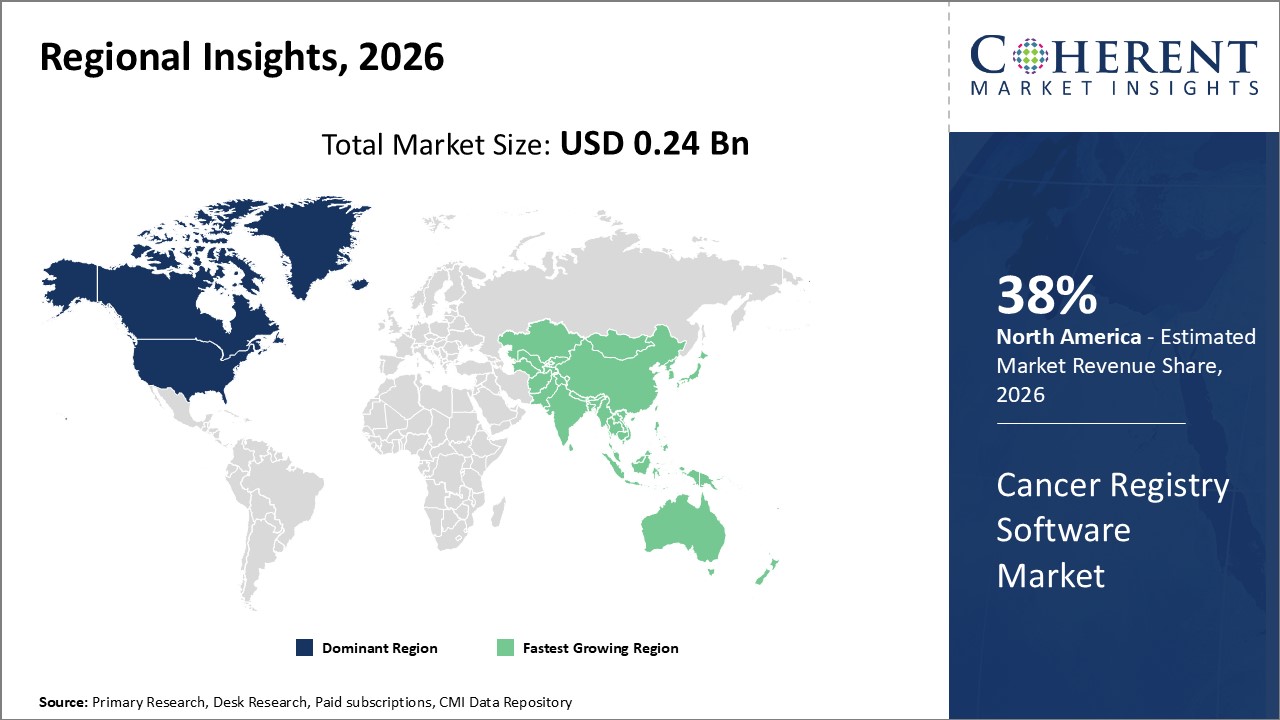
North America leads with strong digital health systems and strict oncology regulations. Asia Pacific grows fastest with rising cancer cases and IT spending. The U.S. advances AI-driven registries, while India scales affordable cloud-based solutions
Cancer Registry Software Market Segmentation Analysis
To learn more about this report, Request Free Sample
Cancer Registry Software Market Insights, By Software Type
The Web-Based segment is the dominating category, capturing 47% of the Cancer Registry Software market due to its scalability, real-time data entry, and seamless integration with hospital IT systems. Cloud-Based solutions represent the fastest-growing segment, supported by flexible access and cost efficiency. On-Premises models are gradually declining due to high upfront costs, while Hybrid and “Others” solutions cater to organizations needing tailored or mixed deployment options.
Cancer Registry Software Market Insights, By End-User
The End User segmentation includes Hospitals, Research Institutions, Government Agencies, Pharmaceuticals & Biotech Companies, and Others. Hospitals dominate the market due to high patient data volumes and strict compliance requirements that drive adoption of advanced cancer registry solutions. Research Institutions are the fastest-growing users, supported by rising oncology research funding and multi-center study needs. Government Agencies rely on registries for surveillance, while Pharma and Biotech firms use them for clinical development. The Others category includes NGOs and smaller providers.
Cancer Registry Software Market Insights, By Functionality
The dominating subsegment is Data Collection & Management, and the fast-growing subsegment is Analytics & Visualization. Regarding Functionality, the market includes Data Collection & Management, Reporting & Compliance, Analytics & Visualization, Patient Tracking, and Others. Data Collection & Management leads due to its role in ensuring accurate, timely, and standardized cancer data. Analytics & Visualization grows rapidly with AI-driven insights. Reporting & Compliance supports regulations, Patient Tracking aids survivorship, and Others cover specialized modules.
Cancer Registry Software Market Trends
The Cancer Registry Software market is rapidly moving toward cloud-based platforms as healthcare systems seek greater flexibility, scalability, and remote accessibility to support modern digital workflows.
A major North American hospital network migrated 80% of its registry infrastructure to the cloud in 2026, achieving a 30% reduction in IT costs, showcasing the financial and operational value driving broader cloud adoption.
AI-powered tools are becoming essential, enabling early cancer detection modeling, predictive risk scoring, and personalized treatment insights that significantly enhance clinical decision-making.
Vendors are expanding open-source modules and interoperability features, improving access to high-quality cancer data for researchers and public health organizations.
The market is evolving from traditional data storage toward real-time, intelligent ecosystems that optimize reporting, analytics, and care coordination.
Cancer Registry Software Market Insights, By Geography
To learn more about this report, Request Free Sample
North America Cancer Registry Software Market Analysis and Trends
In North America, the dominance in the Cancer Registry Software industry stems from a well-established healthcare ecosystem, high adoption of digital health infrastructure, and stringent governmental regulations around oncology data management. The U.S. contributes a significant market share exceeding 30%, reinforced by the presence of key industry players and ongoing innovation in AI-based registry functionalities.
Asia Pacific Cancer Registry Software Market Analysis and Trends
Meanwhile, the Asia Pacific exhibits the fastest growth, driven by rising cancer incidence, expanding healthcare IT budgets, and increasing government cancer control initiatives across India, China, and Southeast Asia. The region's CAGR surpasses other regions due to burgeoning demand for scalable cloud-based registry solutions and growing oncology research collaborations supported by technology vendors.
Cancer Registry Software Market Outlook for Key Countries
USA Cancer Registry Software Market Analysis and Trends
The USA’s Cancer Registry Software market is characterized by rapid modernization and innovation integration. Governmental programs like the National Program of Cancer Registries (NPCR) have driven widespread registry software upgrades, contributing a substantial portion of market revenue. Leading companies, such as IBM Watson Health and Cerner Corporation, have rolled out AI-enhanced registry tools targeting large hospital networks, exemplified by a reported 18% improvement in data accuracy in 2024. The country’s robust research infrastructure and healthcare digitization efforts create sustained demand for advanced registry solutions.
Japan Cancer Registry Software Market Analysis and Trends
India’s market is witnessing accelerated growth due to increased cancer awareness and government initiatives such as the National Cancer Registry Programme (NCRP). Domestic healthcare providers are adopting cost-effective and cloud-based cancer registry software, facilitating better patient tracking across urban and rural settings. Companies collaborating with Indian government health agencies report a 25% increase in cancer data reporting efficiency in 2026. Market players focusing on affordable and scalable solutions are gaining traction in this expansive and underserved market, which forecasts robust business growth despite infrastructural challenges.
Analyst Opinion
Enhanced Data Integration Capacities: Strong interoperability with EHR and hospital information systems is accelerating market growth. In 2024, over 71% of new Cancer Registry Software deployments offered seamless EHR integration, enabling accurate patient data consolidation and real-time updates. This supply-side advancement is directly expanding market share as healthcare institutions prioritize efficiency and compliance.
Regulatory Compliance and Reporting Demand: Global mandates and accreditation requirements continue to push adoption of comprehensive registry solutions. The UK’s NCRAS improved reporting timeliness by 26% in 2024 through upgraded registry tools, demonstrating how demand-side pressures are driving the need for scalable software with advanced reporting capabilities.
Advancements in AI and Analytics Modules: AI-powered predictive analytics and visualization tools are strengthening decision-making. In 2026, U.S. clinical registries achieved a 16% reduction in data processing time due to AI integration, highlighting rising opportunities in data-driven cancer management.
Growing Use Cases in Oncology Research and Public Health: Registries now support broader applications such as public health surveillance and large-scale oncology research. A 2024 multi-institutional study used registry data to assess survival outcomes, emphasizing the software’s scalability and driving global adoption.
Market Scope
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2026 | Market Size in 2026: | USD 0.24 billion |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 9.6% | 2033 Value Projection: | USD 0.46 billion |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
GE Healthcare, IBM Watson Health, Cerner Corporation, Optum, Flatiron Health, Meditech, Epic Systems, Siemens Healthineers, Allscripts, Philips Healthcare, McKesson, OncoHealth, M*Modal, SAS Institute Inc., 3M Health Information Systems, BioFortisTop of Form Bottom of Form |
||
| Growth Drivers: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Cancer Registry Software Market Growth Factors
The Cancer Registry Software market is expanding due to the rising global cancer incidence, which is pushing healthcare providers to adopt more advanced registry systems for accurate patient management and epidemiological monitoring. With the International Agency for Research on Cancer (IARC) reporting 19 million new cancer cases in 2024, the urgency for precise and scalable data solutions has intensified. Government initiatives in countries like the U.S., Canada, and Germany promoting standardized reporting and mandatory cancer surveillance are further accelerating software adoption.
Additionally, the growing shift toward value-based care models in oncology requires detailed tracking of treatment outcomes and survivorship, increasing demand for analytics-driven registry tools. Technological advancements such as cloud computing and AI are also improving software scalability, usability, and accessibility, making modern cancer registry solutions more efficient and easier to deploy across diverse healthcare environments.
Cancer Registry Software Market Development
In March 2026, Paige, a digital pathology startup, launched Paige Breast Lymph Node, AI-powered software designed to help pathologists detect breast cancer that has spread to lymph nodes, including micrometastases. Using its proprietary TissueMap, the software highlights suspicious areas, improving diagnostic efficiency and accuracy. Currently available for research use in the U.S., Paige is pursuing regulatory approvals in the U.S., EU, and U.K., complementing its FDA-authorized Paige Prostate platform.
In November 2026, Savista has acquired ONCO’s cancer registry services division, enhancing its leadership in oncology data management and revenue cycle management (RCM). While ONCO continues to operate its software platforms—including ONCOLog, ONCONav, and OWA—Savista will expand service delivery, AI-driven case finding, and abstraction workflows. The partnership strengthens operational efficiency, compliance, and analytics capabilities, providing scalable, high-quality cancer registry solutions for healthcare providers across the U.S., while ONCO focuses on advancing its innovative cancer registry software.
Key Players
Leading Companies of the Market
GE Healthcare
IBM Watson Health
Cerner Corporation
Optum
Flatiron Health
Meditech
Epic Systems
Siemens Healthineers
Allscripts
Philips Healthcare
McKesson
OncoHealth
M*Modal
3M Health Information Systems
BioFortis
Several leading companies have emphasized strategic collaborations and technology acquisitions to stay competitive. For example, a 2026 joint venture between a major technology provider and a healthcare analytics firm resulted in the launch of an AI-powered oncology registry platform, increasing market presence and expanding clientele in North America. Similarly, strategic partnerships focusing on cloud migration enabled rapid deployment of cancer registry solutions in emerging Asia Pacific markets, accelerating business growth and customer base diversification.
Cancer Registry Software Market Future Outlook
The future outlook for the Cancer Registry Software market remains highly positive, driven by rising digital transformation across healthcare systems and increasing global focus on cancer surveillance. Advancements in AI, machine learning, and predictive analytics will enhance registry capabilities, enabling more accurate risk modeling and personalized treatment insights. Cloud-based platforms will continue gaining traction due to scalability and remote accessibility. Governments are expected to strengthen reporting mandates, further boosting adoption. Additionally, integration with EHRs, real-time data ecosystems, and expanding use in oncology research will position cancer registry software as a critical tool for improving outcomes and shaping public health strategies.
Cancer Registry Software Market Historical Analysis
The Cancer Registry Software market has progressed from basic, manual data entry tools to advanced digital platforms driven by rising cancer incidence and the need for standardized reporting. Earlier systems were mostly on-premises with limited integration and minimal analytics. As electronic health record (EHR) adoption expanded, healthcare institutions sought automated and interoperable registries to improve data accuracy and compliance. From the mid-2010s onward, government mandates in the U.S., Europe, and Asia accelerated adoption. By the early 2020s, advancements in cloud computing, AI-driven analytics, and visualization tools transformed registries into scalable, intelligent systems supporting research and outcome monitoring.
Sources
Primary Research Interviews:
Oncology Data Managers
Cancer Registrars (CTR Professionals)
Hospital IT Directors
Health Information Management (HIM) Specialists
Medical Oncologists & Radiation Oncologists
Databases:
SEER (Surveillance, Epidemiology, and End Results Program)
National Program of Cancer Registries (NPCR)
WHO Global Cancer Observatory (GLOBOCAN)
ClinicalTrials.gov
Global Health Data Exchange (GHDx)
Magazines:
Healthcare IT News
Health Data Management
Modern Healthcare
Oncology Times
Journals:
Journal of Registry Management
Cancer Epidemiology, Biomarkers & Prevention
Journal of the American Medical Informatics Association (JAMIA)
International Journal of Medical Informatics
Associations:
North American Association of Central Cancer Registries (NAACCR)
American Health Information Management Association (AHIMA)
International Association of Cancer Registries (IACR)
American Society of Clinical Oncology (ASCO)
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients