Chordoma Disease Therapeutics Market is estimated to be valued at USD 7,378.9 Mn in 2026 and is expected to reach USD 11,926.6 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 7.1% from 2026 to 2033.
Analysts’ Views on Global Chordoma Disease Therapeutics Market:
Increasing prevalence of and awareness about chordoma disease, rising geratic population, and new drugs releases and strategic efforts by prominent market competitors are favorably impacting the market's growth. The high prevalence of cancer, favorable health reimbursement, and increased awareness regarding cancer among people and clinicians is a major factor in the chordoma disease therapeutics market, thereby contributing to the market growth.
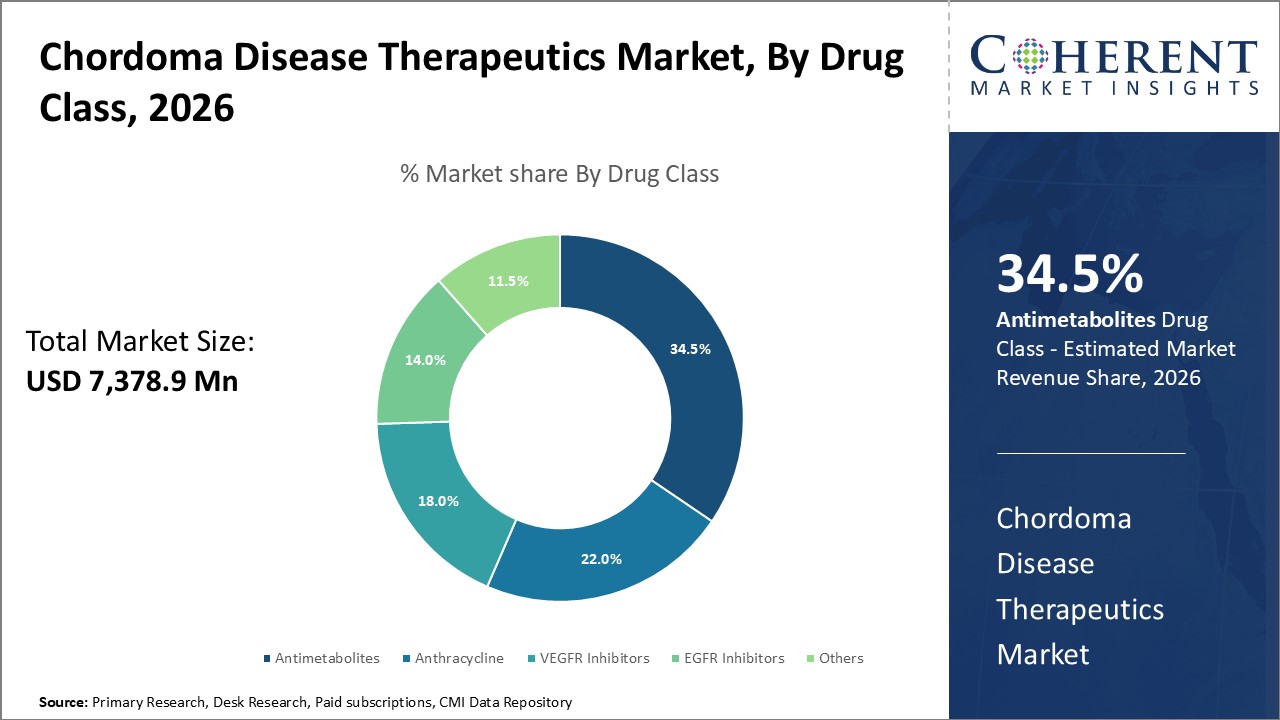
Figure 1. Global Chordoma Disease Therapeutics Market Share (%), By Drug Class, 2026
To learn more about this report, Request Free Sample
Global Chordoma Disease Therapeutics Market– Driver
Increasing prevalence of chordoma cancer
Increasing prevalence of chordoma cancer is expected to propel growth of the global chordoma disease therapeutics market over the forecast period. For instance, according to an article published in the World Health Organization, in February 2022, the age-standardized point prevalence and annual incidence rates of Cancer were 246.6 and 14.9 in 2021, which increased by 7.4% and 8.2% from 1990, respectively. Cancer is a leading cause of death worldwide, accounting for nearly 10 million deaths in 2020. As per the national orgniasation of rare disorders reported, the incidence of chordoma is estimated approximately 1 per 1,000,000 people. About 300 cases of chordoma are diagnosed in the U.S. each year.
Increasing drug approvals for chordoma treatment
Increasing drug approvals from regulatory authorities for the treatment of chordoma is expected to drive growth of the chordoma disease therapeutics market over the forecast period. For instance, in December 2022, MaxiVAX, a biotechnology company, developed a cancer vaccine comprising an immune boost agent released in situ by an engineered cell line and cancer autologous cells providing antigens. A Phase I trial with 25 patients with an advanced stage of cancer showed encouraging efficacy data and safety profile of the treatment. The European union -funded MaxONCO project Phase II clinical trial evaluate this vaccine in 45 patients with head and neck cancer and in chordoma patients. The chordoma trial has been conducted in the EU and USA as an Investigational New Drug Programme that allows fast-track marketing of the drug in case of successful trial results.
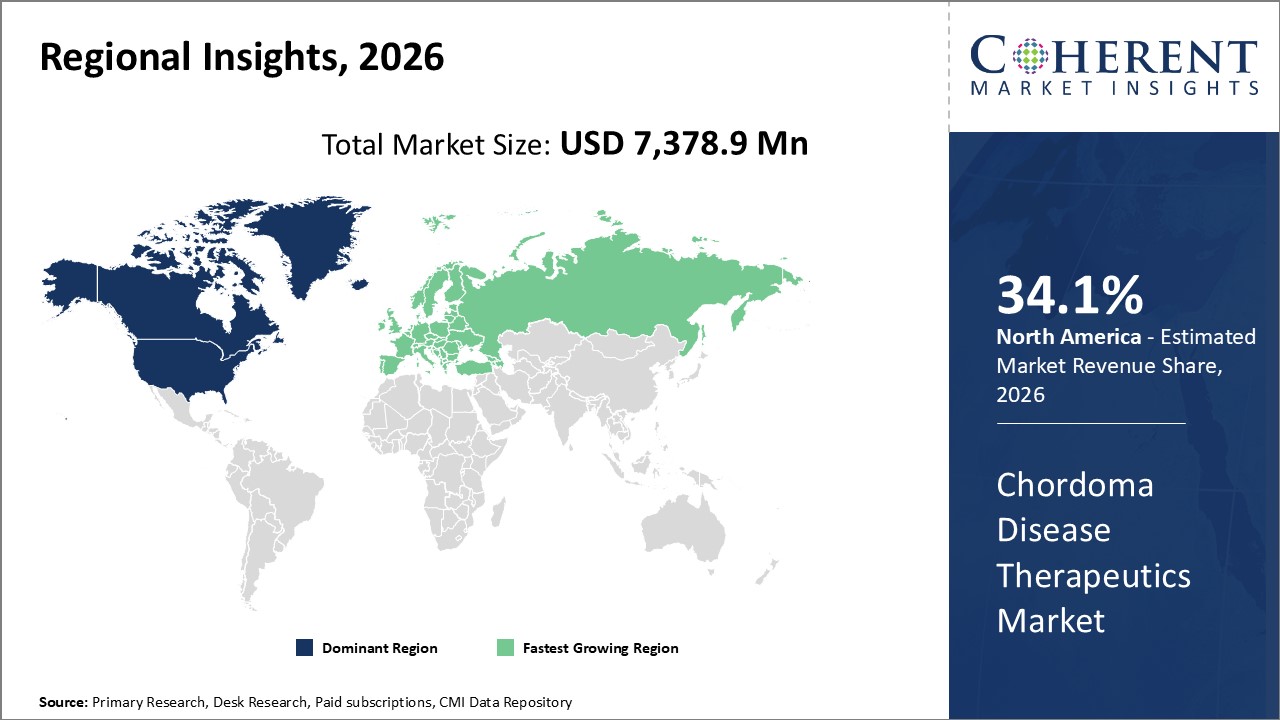
Figure 2. Global Chordoma Disease Therapeutics Market Value (US$ Million), by Region, 2026
To learn more about this report, Request Free Sample
Global Chordoma Disease Therapeutics Market- Regional Analysis
Among regions, North America is estimated to hold a dominant position in the global chordoma disease therapeutics market over the forecast period. North America is estimated to hold 34.1% of the market share in 2026. The global chordoma disease therapeutics market is expected to witness significant growth in the coming years, driven by the high prevalence of cancer, favorable health reimbursement, and increased awareness, North America to emerge as the leading region for the chordoma disease therapeutics market. For instance, according to the press release in February 2020, by imaware, provides laboratory testing for wellness monitoring, informational, and educational use in North America, announced the launch of the At-Home Cancer Screening Test, designed by healthcare company Micro drop. The imaware test for cancer uses just a few drops of blood to detect unique biomarkers as well as two additional drugs to provide more comprehensive results. These kinds of developments in the healthcare system and rising health expenditures, as well as increased knowledge of advanced cancer treatments, are fueling the chordoma disease therapeutics market in North America.
Even though online consultations are advisable with patients who are having stable cancer.
Chordoma Disease Therapeutics Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 7,378.9 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.1% | 2033 Value Projection: | USD 11,926.6 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
GlaxoSmithKline Plc, Bavarian Nordic A/S, Boehringer Ingelheim International GmbH, Pfizer, Inc., Merck & Co., Inc., Sanofi SA, Astellas Pharma Inc., AstraZeneca plc, Amgen, Inc., Bristol-Myers Squibb and Company, Novartis International AG, Bayer AG, Dr. Reddy’s Laboratories Ltd., Celgene Corporation, Eli Lilly and Company ayer AG, Johnson & Johnson Services, Inc , Merck & Co., Inc., Optivus Proton Therapy, Inc, ProCure Treatment Centers, Inc., Varian Medical Systems, Inc. Amura Holdings Ltd., Catena pharmaceuticals Inc., Celldex Therapeutics , Eckert & Ziegler BEBIG, Infinity Pharmaceuticals, Inc, Medivir, Verdict Media Limited, Mylan N.V. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Chordoma Disease Therapeutics Market Segmentation:
The Global chordoma disease therapeutics market report is segmented By Drug Class, By Treatment Type, By End User and Region.
By Drug Class, the market is segmented into antimetabolites, anthracycline, vegfr inhibitors, EGFR inhibitor, and others. Out of which, the antimetabolites is expected to hold a dominant position in the global chordoma disease therapeutics market during the forecast period and this is attributed to awareness about chordoma disease.
By Treatment Type, the market is segmented into chemotherapy, targeted therapy, radiation therapy, and others. Out of which, the chemotherapy is expected to dominate the market over the forecast period and this is attributed to prompt treatment can lead to proper recovery.
By End User, the market is segmented into hospitals, clinics, oncology centers, and others. Out of which, hospitals is expected to dominate the market over the forecast period and this is attributed to prevention, diagnosis, multidisciplinary treatment, supportive care, research and education.
Among all the segmentations, the treatment type segment expected to dominate the market over the forecast period and this is attributed to the block tumour growth in chordoma disease is which is expected boost the growth of chordoma disease therapeutics market over the forecast period.
Global Chordoma Disease Therapeutics Market Cross Sectional Analysis:
Key players are making chordoma disease therapeutics with proper drug treatment emerging economies is also expected to boost demand chordoma disease therapeutics market in North America region. For instance, in October 2020, Myriad Genetic Laboratories, Inc., a genetic testing and precision medicine company based in Salt Lake City, Utah, U.S., introduced Myriad's EGFR inhibitor treatment method from the Japanese Ministry of Health, Labor, and Welfare to be utilized as a partnered treatment.
Global Chordoma Disease Therapeutics Market: Key Developments
On May 31, 2023, Boehringer Ingelheim International, a pharmaceutical company, developed Afatinib dimaleate, a 4-anilinoquinazoline, antineoplastic agent. It is formulated as film coated tablets for oral route of administration. It is indicated for the first-line treatment of patients with metastatic non-small cell lung cancer (NSCLC) tumors have epidermal growth factor receptor (EGFR) exon-19 deletions or exon-21 (L858R) substitution mutations as detected by an US FDA-approved test, indicated as monotherapy for the treatment of Epidermal Growth Factor Receptor (EGFR) tyrosine kinase inhibitor naïve patients with metastatic (including cytologically proven pleural effusion) adenocarcinoma of the lung with activating EGFR mutation(s).
In December 2020, Bavarian Nordic A/S, a integrated vaccine company focused on the development, manufacturing and commercialization of life-saving vaccines in Denmark, announced top-line results from a Phase 2 clinical study evaluating the targeted immunotherapy candidate, BN-Brachyury in the treatment of advanced chordoma, a rare cancer occurring in the base of the skull and spine. While the study failed to meet its primary endpoint, it provided evidence of clinical activity.
On June 1, 2023, Myriad Genetics, Inc., is a company of genetic testing and precision medicine, launched new studies and expansion of its Precise Oncology Solutions portfolio at the 2023 American Society of Clinical Oncology (ASCO).
On March 22, 2023, F. Hoffmann-La Roche Ltd (Roche), a biotechnology company that develops drugs and diagnostics to treat major diseases, announced that it has entered into a collaboration with Eli Lilly and Company is pharmaceutical company headquartered in U.S. to support the development of Roche’s Elecsys chordoma disease therapeutics.
In March 2022, Invitae, a medical genetics company, announced full access to its Personalized Cancer Monitoring (PCMTM) platform to help detect minimal or molecular residual disease (MRD) in patients with solid tumors. Invitae PCM uses a novel set of personalized assays based on a patient's tumor to detect circulating tumor DNA (ctDNA) in blood, offering the ability to perform risk stratification, response assessment to treatment and detection of cancer recurrence, based on recent studies.
Global Chordoma Disease Therapeutics Market: Key Trends
Application of biomarker test for diagnosing chordoma disease
Application of biomarker test for diagnosing chordoma disease is expected to propel growth of the global chordoma disease therapeutics market over the forecast period. Major players in the market are focused on adopting partnership strategies to expand its product portfolio. Presently, there is no drugs available for the diagnosis of chordoma disease. However, anti–cyclic citrullinated peptide (anti–CCP) antibodies test require more refinement to improve its clinical utility. For instance, in March 2020, WorldCare Clinical, LLC, an independent contract research organization offering imaging in clinical trials, partnered with Navidea Biopharmaceuticals, Inc., a biopharmaceutical company focused on the development of precision immunodiagnostic agents and immuno-therapeutics, to offer imaging service following U.S. FDA approval of Navidea’s cancer diagnostic. Navidea’s strategy is to deliver novel products and advancing the company’s pipeline through global partnering with WorldCare Clinical, LLC.
Global Chordoma Disease Therapeutics Market: Restraint
Chemotherapeutic drugs approved for the treatment of chordoma are Showing side effects
Chemotherapeutic drugs approved for the treatment of chordoma are not always effective and have several side effects such as nausea, headache, and hair loss is expected to hamper the growth of the global chordoma disease therapeutics market. For instance, according to an article published in the National library of medicine, on February 27, 2023, reported antimetabolites have a sevral side effects such as dose-limiting myelosuppression. Toxic levels of 5-fluorouracil occur in patients with Dihydropyrimidine Dehydrogenase (DPD) deficiency or drug overdose. This lead to cardiac dysfunction, colitis, neutropenia, and encephalopathy.
Key market Players are focusing on Safety and efficacy of drugs.
Global Chordoma Disease Therapeutics Market- Key Players
Major players operating in the global chordoma disease therapeutics market include GlaxoSmithKline Plc, Bavarian Nordic A/S, Boehringer Ingelheim International GmbH, Pfizer, Inc., Merck & Co., Inc., Sanofi SA, Astellas Pharma Inc., AstraZeneca plc, Amgen, Inc., Bristol-Myers Squibb and Company, Novartis International AG, Bayer AG, Dr. Reddy’s Laboratories Ltd., Celgene Corporation, Eli Lilly and Company ayer AG, Johnson & Johnson Services, Inc , Merck & Co., Inc., Optivus Proton Therapy, Inc, ProCure Treatment Centers, Inc., Varian Medical Systems, Inc. Amura Holdings Ltd., Catena pharmaceuticals Inc., Celldex Therapeutics, Eckert & Ziegler BEBIG, Infinity Pharmaceuticals, Inc, Medivir, Verdict Media Limited, and Mylan N.V.
Global Chordoma Disease Therapeutics Market– Definition
Chordoma is a rare type of cancer, which occurs in the spine region or from the base of the skull to the tailbone. Chordoma is included in a group of malignant bone and soft tissue tumors called sarcomas. Chordomas are the most common tumor of the sacrum and cervical spine. Chordoma tumors can press the spine, brain, and nerves as they grow causing nerve problems such as tingling, numbness, weakness, lack of bladder or bowel control, sexual dysfunction, vision problems, endocrine problems, and swallowing difficulties.
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients