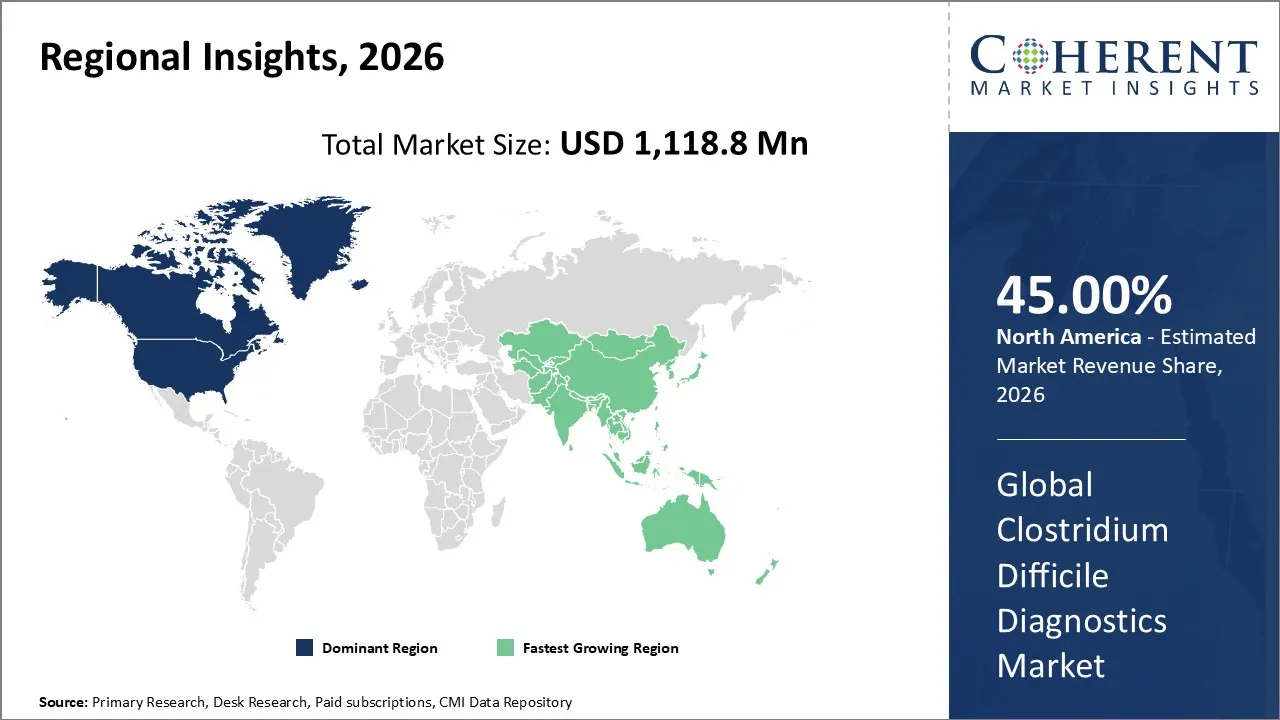
The global clostridium difficile diagnostics market size is expected to reach approximately USD 1,118.8 Mn in 2026 and USD 1,852.1 Mn by 2033, growing at a CAGR of 7.5% throughout the forecast period (2026-2033).
The laboratory stool tests used to detect toxin-producing bacteria that cause colitis and diarrhea, typically following antibiotic use, are known as Clostridioides difficile (C. diff) diagnostics. In individuals whose normal gut microbiota has been altered by chemotherapeutic or antibiotic treatments, Clostridium difficile, an anaerobic, gram-positive bacterium, causes serious illness by generating two powerful toxins (dysbiosis). According to the guidelines released by the regulatory bodies a two-step procedure is recommended, starting with a glutamate dehydrogenase (GDH) test and ending with a toxin or nucleic acid test. Alternatively, a nucleic acid test by itself is appropriate in establishments where testing is guided by recognized clinical algorithms. The clostridium difficile diagnostic market is gaining strategic importance in the market due to increasing prevalence of clostridium difficile infection, launch and approval of new product and technology advancement.
|
Current Event |
Description and its Impact |
|
Growing Adoption of Molecular Diagnostic Technologies |
|
|
Rising Prevalence of Hospital-acquired infections (HAIs) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Based on diagnostic test, enzyme immunoassays are expected to lead the market, accounting for a revenue share of 50% in 2026. The enzyme immunoassays are cheaper than molecular tests and hence used extensively in routine hospital diagnostics. Enzyme immunoassays require less complex infrastructure and are easier to integrate into standard microbiology workflows. Enzyme immunoassays are not only utilized as standalone toxin tests; they are also embedded in commonly recommended clostridium difficile infection testing algorithms such as GDH + toxin EIA, GDH + toxin EIA arbitrated by Nucleic Amplification Tests (NAAT), or NAAT + toxin. That keeps enzyme immunoassays diagnostic test demand high even when labs do not rely on it alone.
By end user, the hospitals segment is slated to account for a prominent market share of 40% in 2026. Hospitals have strong in-house diagnostic infrastructure and well-trained staff for performing diagnosis tests. Hospitals lead the Clostridium difficile diagnostics market due to the presence of high-risk patient volume, availability of advanced diagnostic infrastructure, central role in diagnosis, treatment, and monitoring.
To learn more about this report, Request Free Sample
North America is expected to dominate the global clostridium difficile diagnostics market, accounting for a share of 45% in 2026. Increasing prevalence of clostridium difficile infection in the North America region has driven the growth of the market. Due to high disease burden in the region, the demand for clostridium difficile diagnostics has also increased. For instance, in March 2026, according to the data published by the U.S. Centers for Disease Control and Prevention, it has been estimated that North America (NA) has the highest reported incidence of Clostridium difficile per 1000 admissions (4.85) and per 10,000 patient-days (6.23).
Another factor driving the growth of the clostridium difficile diagnostics market in North America is early adoption of molecular and guideline-based testing algorithms. North America also benefits from a strong regulatory and commercial ecosystem. The U.S. FDA has cleared or referenced multiple CDI-related diagnostic approaches in regulated clinical settings, and the region has a dense network of large hospital systems, CLIA-certified labs, and diagnostic manufacturers/distributors. So, the growth of the market in North America has been boosted by having advanced lab facilities, support for payments, and the widespread use of NAAT/PCR and multistep testing methods.
Asia Pacific is expected to grow at the fastest CAGR in the clostridium difficile diagnostics market during the forecast period. Counteries like China, India, and Japan are heavily investing in hospitals, laboratories, and diagnostic centers. Asia Pacific has a huge patient pool, especially elderly individuals who are more prone to infections like clostridium difficile. Rising hospitalization rates and antibiotic exposure are increasing clostridium difficile infection cases, which has estimated to drive the need for faster and accurate diagnostic testing in the region. Increasing government initiatives in Asia Pacific region and growing awareness campaigns has driven the market growth. Overall, Asia Pacific is growing at significant rate due to its large population, rising infection rates, improving healthcare systems, and increasing adoption of advanced diagnostics.
The U.S. country held majority revenue share in the North America clostridium difficile diagnostics market. The U.S. has one of the highest healthcare expenditures globally, which enables adoption of expensive but accurate diagnostic tools. For instance, in February 2026, according to the data published by the Institute for Health Metrics and Evaluation, when measured from birth, improvements in medical treatment extended health spans in the U.S. by 1.3 years and raised medical spending by USD 234,000 per person over their lifetime, or almost USD 182,000 every extra year of healthy life acquired. These are some of the main conclusions of a recent comprehensive nationwide study that was just released in Value in Health. A major reason is the high volume of hospital-based and healthcare-associated testing in the U.S. clostridium difficile diagnostics is closely tied to hospitalization, antibiotic exposure, and infection-control programs, and the U.S. has a very large hospital network and a mature surveillance ecosystem. Additionally, the U.S. leads due to the faster adoption of high-end diagnostic technology, particularly FDA-approved PCR/NAAT-based diagnostics.
China leads the Asia Pacific clostridium difficile diagnostics market due to large patient pool and rapid expansion of healthcare infrastructure. The Chinese government is actively investing in infection control programs, hospital upgrades, and diagnostic technology adoption. China country is increasingly adopting molecular diagnostics (NAAT, PCR) and rapid diagnostic kits. Hospitals are integrating modern diagnostic platforms to improve accuracy and turnaround time.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1,118.8 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.5% | 2033 Value Projection: | USD 1,852.1 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
F. Hoffmann-La Roche Ltd, Thermo Fisher Scientific, Merck & Co., Inc., Pfizer, Inc., Actelion Pharmaceuticals, Inc., Abbott Laboratories, Trinity Biotech Plc., Summit Therapeutics, Sanofi S.A., Novartis AG, and AstraZeneca Plc |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Rising prevalence of clostridium difficile infection cases globally has directly increased the volume of diagnostic tests performed in labs and hospitals. Studies show that increasing prevalence of clostridium infections creates a strong demand for accurate and timely diagnostics. A large proportion of clostridium difficile diagnostics cases are hospital-acquired infections (HAIs). This leads hospitals to implement routine screening protocols and invest in advanced diagnostic systems. Due to overuse of antibiotics disrupts gut microbiota which causes clostridium difficile infection risk. Rising prevalence of clostridium difficile infection in elderly population, immunocompromised patients and long-term hospitalized individuals increases screening frequency and demand for routine diagnostic monitoring.
Rising approval of diagnostic tests/ assays by regulatory authority’s bodies like U.S. FDA, CE, CDSCO, etc.) has driven the growth of the clostridium difficile diagnostics market. Regulatory approvals allow faster commercialization & market entry which accelerates the growth of the market. Regulatory bodies ensure analytical and clinical validation of diagnostic tests before approval which guarantees sensitivity, reliability and accuracy, which is critical for infectious diseases like clostridium difficile. Strong regulatory frameworks ensure that only high-quality diagnostic tools reach the market, eliminating low-performance tests. Physicians are more confident in prescribing approved tests hence the increasing regulatory approval of diagnostics test/ assays is driving the growth of the market.
Multiplex GI panels can deliver broad results more quickly than conventional multi-step testing workflows; they help clinicians identify infectious causes sooner and act faster on clostridium difficile diagnostics related treatment and infection-control measures. Using multiplex GI panels give better antimicrobial stewardship and workflow efficiency. As the multiplex GI panels supports faster diagnosis by enabling simultaneous detection of clostridium difficile and other enteric pathogens from a single sample, reducing diagnostic turnaround time, improving clinical decision-making there is rise in the adoption of the multiplex GI panels for diagnosis.
Strong demand for the tied to antimicrobial stewardship has created significant opportunity for the growth of the clostridium difficile diagnostics market in the near future. Clostridium difficile infection diagnostics become more crucial when healthcare systems prioritize cutting down on unnecessary antibiotic use because quicker and more accurate confirmation enables more focused therapy. This increases the clinical and financial value of clostridium difficile infection testing in hospital decision-making.
Share
Share
Abhijeet Kale is a results-driven management consultant with five years of specialized experience in the biotech and clinical diagnostics sectors. With a strong background in scientific research and business strategy, Abhijeet helps organizations identify potential revenue pockets, and in turn helping clients with market entry strategies. He assists clients in developing robust strategies for navigating FDA and EMA requirements.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients