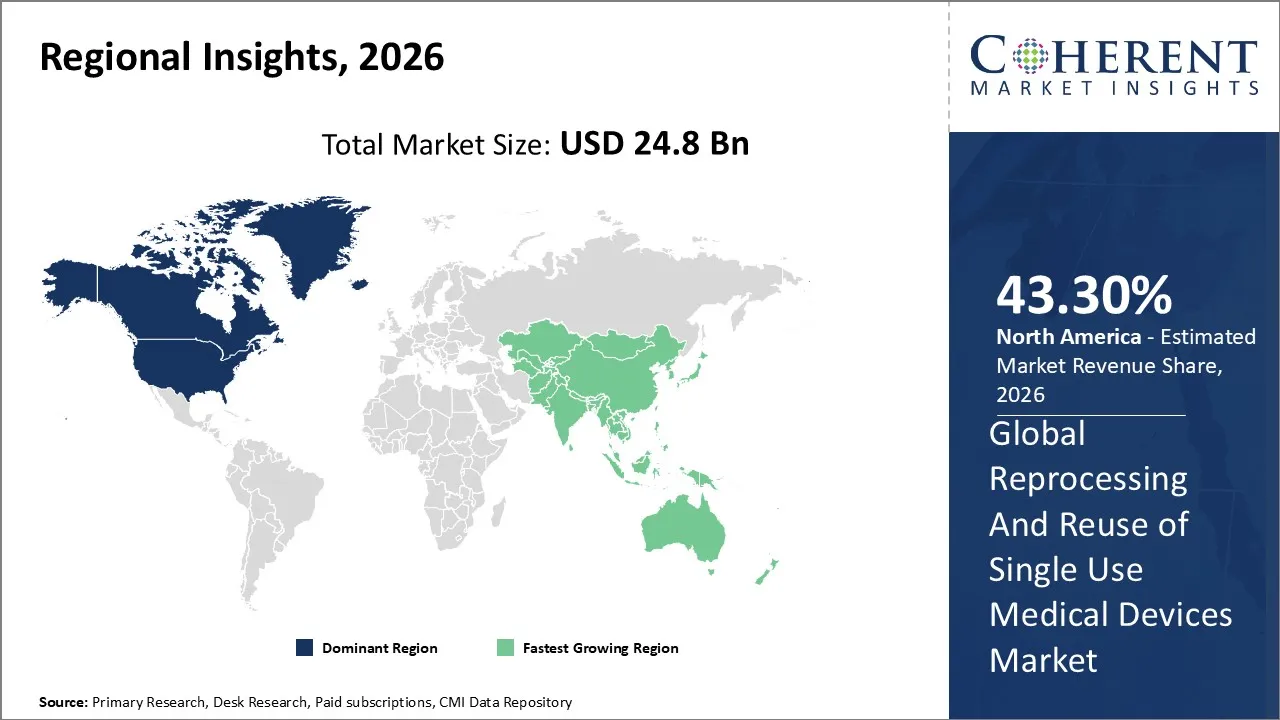
The Reprocessing and Reuse of Single Use Medical Devices Market is estimated to be valued at USD 24.8 Bn in 2026 and is expected to reach USD 36.7 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 11.3% from 2026 to 2033.
The Reprocessing and Reuse of Single-Use Medical Devices Market involves organizations collecting, cleaning, sterilizing, testing, and re-certifying devices originally labeled for single use to enable their safe reuse. Rising healthcare costs, increasing surgical procedures, and stronger sustainability goals drive market growth. Hospitals and ambulatory surgical centers actively adopt reprocessed devices, supported by stringent regulatory frameworks and advanced sterilization technologies. North America drives adoption, while Europe and Asia-Pacific expand their presence by prioritizing cost efficiency and environmental sustainability initiatives.
|
Current Events |
Description and its impact |
|
Regulatory Changes in Key Regions |
|
|
Technological Advancements in Sterilization and Tracking |
|
|
Public Health Trends and Infection Control Priorities |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Hospitals/Clinics acquired the prominent market share of 42.2% in 2026. Hospitals and clinics actively fuel the growth of the Reprocessing and Reuse of Single-Use Medical Devices Market by adopting reprocessed instruments to control healthcare costs and enhance operational efficiency. High patient volumes and frequent surgeries generate steady demand for reusable devices. Established sterile processing departments and partnerships with certified third-party reprocessors allow healthcare facilities to reuse devices safely and reliably. Furthermore, sustainability initiatives and strict regulatory compliance motivate hospitals and clinics to implement reprocessing programs, cut medical waste, and deliver cost-effective, high-quality patient care.
Class II hold the largest market share of 52.3% in 2026. Class II devices significantly propel the Reprocessing and Reuse of Single-Use Medical Devices Market because healthcare providers widely use them in complex procedures and purchase them at relatively high costs. Hospitals actively reprocess these moderate-risk devices to lower procurement spending while maintaining strong clinical performance. Advanced cleaning validation and functional testing technologies enable safe and reliable reuse. Clear regulatory guidelines and structured approval pathways further motivate healthcare facilities to incorporate Class II device reprocessing into their standard operational practices. For instance, in June 2025, Cardinal Health launched a U.S. multi-parameter monitoring system that continuously tracks cardiac activity, blood oxygen, and temperature through a single patient-use cable and lead wire.
Cardiology expected to hold largest market share of 35.6% in 2026. Cardiology strongly propels the Reprocessing and Reuse of Single-Use Medical Devices Market as healthcare providers perform a high number of interventional procedures and rely heavily on advanced disposable devices such as electrophysiology catheters and diagnostic mapping tools. The rising prevalence of cardiovascular diseases pushes hospitals to seek cost-effective treatment options, leading them to adopt reprocessed devices. Robust quality assurance measures and demonstrated clinical effectiveness build physician trust. Efforts to control catheterization lab costs and minimize procedural waste further drive reprocessing adoption in cardiology units.
To learn more about this report, Request Free Sample
North America dominates the overall market with an estimated share of 43.3% in 2026. In North America, hospitals and surgical centers actively drive the Reprocessing and Reuse of Single-Use Medical Devices Market by adopting cost-saving strategies and reprocessing high-value devices to control budgets and minimize waste. Providers leverage advanced sterilization technologies, regulatory guidance, and established third-party reprocessing services to ensure safe and efficient operations. Sustainability efforts and environmental regulations further motivate healthcare facilities to implement reuse programs, while growing procedural volumes and ongoing technological innovations continue to expand the market throughout the United States and Canada. For instance, in May 2024, Cook Medical launched its Ascend Single-Use Flexible Ureteroscope in the U.S. and Canada, expanding its stone management offerings for urology customers.
Hospitals and clinics in the Asia Pacific region actively drive the growth of the Reprocessing and Reuse of Single-Use Medical Devices Market by adopting cost-effective solutions to address increasing healthcare demands. Providers respond to rising medical waste and growing sustainability awareness by implementing reprocessing programs. They invest in advanced sterilization technologies and collaborate with certified reprocessing partners to ensure device safety and reliability. Expanding surgical volumes, higher chronic disease rates, and strengthening regulatory frameworks further propel market growth across countries including China, India, Japan, and Australia.
Hospitals and surgical centers in the United States actively drive the growth of the Reprocessing and Reuse of Single-Use Medical Devices Market by adopting cost-saving strategies to manage high healthcare expenditures. Providers reprocess high-value devices to lower procurement costs while maintaining safety and performance standards. They rely on advanced sterilization technologies, strict FDA regulations, and extensive third-party reprocessing services to ensure reliable reuse. A stronger focus on sustainability and waste reduction, along with increasing procedural volumes and technological advancements, further propels market expansion across healthcare facilities nationwide. For instance, in December 2025, Edgewell Personal Care launched the Schick and Skintimate Free Recycling Program with TerraCycle, letting U.S. consumers recycle all disposable razors.
Hospitals and clinics across China actively drive the growth of the Reprocessing and Reuse of Single-Use Medical Devices Market by adopting cost-effective strategies to manage increasing patient volumes and rising healthcare needs. Providers implement reprocessing programs to lower procurement costs while maintaining device safety and performance. They invest in advanced sterilization technologies and work closely with certified reprocessing partners to ensure reliability. Government initiatives promoting environmental sustainability, tighter regulatory oversight, and expanding surgical procedures further accelerate the adoption of reprocessed devices in both urban and regional healthcare facilities.
Healthcare facilities increasingly integrate reprocessing programs to manage costs and optimize device utilization. High-value single-use devices, such as catheters and endoscopes, are prioritized for reuse. Hospitals leverage both in-house sterile processing departments and third-party certified reprocessors to maintain safety and efficiency. The trend reflects a shift toward operational cost management, improved workflow efficiency, and sustainable practices, making reprocessing a routine part of hospital procurement strategies while ensuring consistent clinical performance and reliability.
Innovations in sterilization technologies, including automated cleaning systems, advanced sterilizers, and functional testing, enhance the safety and reliability of reprocessed devices. Real-time validation, device tracking, and analytics tools allow healthcare providers to monitor performance and compliance. These technological improvements reduce infection risks and support regulatory requirements, enabling broader adoption of reprocessing programs across clinical departments. Providers increasingly rely on these tools to ensure device functionality and patient safety while minimizing operational bottlenecks.
Reprocessing high-value single-use devices offers hospitals and clinics a strategic opportunity to reduce procurement costs while maintaining clinical outcomes. Facilities can reinvest the savings into expanding services or upgrading infrastructure. End users can implement standardized reprocessing programs and leverage third-party certified providers, ensuring safety and reliability. This opportunity is particularly significant for departments with high procedural volumes, such as cardiology and gastroenterology, where device reuse directly impacts operational efficiency and budget management.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 24.8 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 11.3% | 2033 Value Projection: | USD 36.7 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Stryker Corporation, Medline Industries, Inc., Suretek Medical, Renu Medical Inc., Johnson & Johnson, Northeast Scientific Inc., Medtronic Plc, Steripro Canada, Vanguard AG, Innovative Health, Verathon Inc., and Hogy Medical Co., Ltd. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients