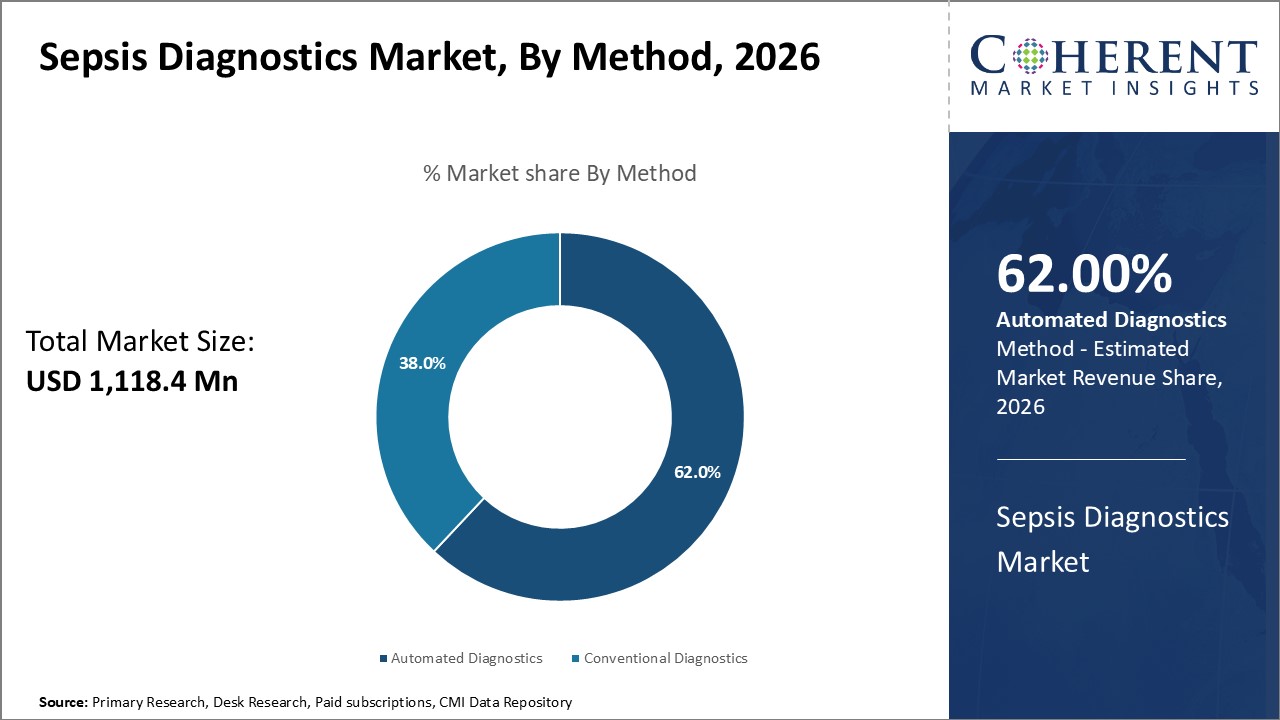
Sepsis Diagnostics Market is estimated to be valued at USD 1,118.4 Mn in 2026 and is expected to reach USD 2,193.3 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 10.1% from 2026 to 2033.
The sepsis diagnostics market is growing steadily, driven by the rising demand for rapid, point-of-care diagnostics and the integration of AI-based early warning systems—factors that support wider adoption of tools like Masimo’s Sepsis Index in clinical settings. For instance, In April 2023, Masimo received FDA clearance for an over-the-counter opioid overdose monitor, expanding its remote monitoring technology to detect respiratory depression at home—similar to how the Sepsis Index identifies early signs of sepsis in hospitals using the Masimo Patient SafetyNet system.
|
Event |
Description and Impact |
|
Artificial Intelligence and Machine Learning Integration in Healthcare |
|
|
Supply Chain Disruptions and Geopolitical Tensions |
|
|
Rising Antimicrobial Resistance (AMR) Crisis |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Significant improvements in the development of biomarker-based assays, molecular technologies, and AI systems for diagnostics are redefining the patent landscape in the sepsis diagnostics market. Immunexpress, Inflammatix, and MeMed Diagnostics are some of the major industry players who obtained precision diagnostics patents, as these companies are now focusing on host-response gene expression assays and multiplex biomarker panels.
Recent patents underscore the importance of Artificial Intelligence and Machine Learning (ML) in improving the sensitivity and specificity of sepsis diagnosis. Cytovale, Inc. for example, patented a multi-omics machine learning sepsis rapid diagnosis platform, which automates the interpretation of intricate biological information.
Other notable examples include the application of nanotechnology and spectroscopy where several universities and startups are working on SERS technologies for prompt pathogen detection without the need for labels. The patent environment also remains contested and includes multiple applications for diagnostic markers Presepsin, CD64 expression, and even new microRNA panels which strengthen the self-sufficient biomarker signatures.
The United States and Europe are still the frontrunners for gaining patent rights as there are stronger legal systems and untapped market value. To enhance and maintain their market competitiveness as well as attract new investments, companies must strategically expand their patent portfolios.
The global sepsis diagnostics market deals with a multi-layered reimbursement setting which greatly affects market coverage and acceptance. The market is currently valued at approximately $685 million and is projected to grow to $1.2 billion by 2030. There is a difference in reimbursement policies in the two regions. In the US, there is CTP billing for sepsis diagnostics. Also, there are two molecular rapid tests which fall under the Accelerate PhenoTest and Verigene GCAP.
For medicare, reimbursement for blood cultures is $25-$45 and for rapid molecular panels $150-$300. Procalcitonin tests are reimbursed at $35-$50. There are some diagnostics which are uncovered, and prior authorization is an issue with private payers. The break through device designation by the FDA speeds up the process. In Europe, reimbursement is supplemented by the SNOMED CT and national systems such as the G-DRG in Germany, T2A in France, and HRG in the UK which pay per case for sepsis (€3,800-8,200).
The IVDR and HTA in the EU set the market entry and target launch windows to 18-36 months. All in all, reimbursement policies are shifting to a focus on encouraging innovation while managing costs, which in turn drives adoption.
A precise, rapid-acting diagnostic system is necessary for the timely decisions concerning the treatment of sepsis. Fast-acting and decisive diagnostic systems are preferred, with results coming in at the exact moment of treatment, at all stages. For early detection of sepsis and SIRS, clinicians utilize blood cultures and procalcitonin tests, accepting a two-hour wait time for results.
For confirmed sepsis with organ dysfunction, the focus shifts to molecular diagnostics and biomarker panels, which must deliver results in under 90 minutes. In these cases, rapid-acting decision making is important in tailoring therapy, which must always be high-urgency.
In the case of septic shock, the focus is put on immediate intervention that is enabled by results from continuous monitoring of relevant biomarkers. For these cases, the turnaround time must be under an hour. In regard to treatment of sepsis, first line therapy is dominated by broad-spectrum antibiotics consisting of Piperacillin-Tazobactam, Meropenem, and Vancomycin. Guided by blood cultures and procalcitonin levels, these are the first relied upon.
When resistance is seen from these, line shifts to second-line positional agents. Second-line is now guided by rapid molecular susceptibility testing and biomarker trends. In the end, diagnostics combining speed and precision with the ability to interact with the treatment plan and the need for proper allocation of antibiotics is the goal for all stages of the disease and outcome.
Innovations in sepsis diagnostics are rapidly advancing, with a strong focus on improving the accuracy and speed of current tests. Several candidates in late-stage clinical trials leverage the role of biomarkers in sepsis diagnostics, such as gene expression assays and biomarker panels, to enhance early-stage sepsis identification. For example, SeptiCyte RAPID and IntelliSep utilize host response gene expression and biomarker panels (IL-6, PCT, CRP) respectively, showing promising results in Phase III trials.
The growing importance of AI in sepsis diagnostics is reflected in early-phase candidates like SeptiScope, a machine learning-based multi-omics platform, and AI-powered sepsis prediction tools analyzing EHR data for automated sepsis detection. These technologies aim to improve diagnostic accuracy and support timely clinical decisions, especially critical in sepsis diagnostics in emergency care settings.
Point-of-care (POC) sepsis diagnostics, such as rapid immunoassays and quantum dot-based panels, offer the potential to reduce turnaround times significantly. Research into sepsis biomarkers continues to expand with novel multiplex panels and spectroscopic techniques in preclinical stages, contributing to more sensitive and specific diagnostic tools.
Overall, the pipeline reflects a strong trend toward integrating automated sepsis diagnostic systems with advanced biomarker research and AI, driving improved early detection and patient outcomes. Regulatory approvals expected in 2024-2025 could accelerate market availability of these cutting-edge diagnostics.
The Instruments segment is projected to hold the dominant position with 30.0% share in the global sepsis diagnostics market in 2026. This is due to the growing rate of positive responses received from regulatory bodies, which leads to the adoption of cutting-edge diagnostic tools. Tools are essential to improving the precision and promptness of sepsis diagnosis which is needed to initiate treatment.
Other significant segments such as Blood Culture Media, Assays & Reagents, and Software also contribute meaningfully to the market landscape. Blood Culture Media is vital for isolating and identifying sepsis-causing pathogens, while Assays & Reagents support the rapid and specific detection of biomarkers. Software solutions are increasingly integrated to improve data management and diagnostic workflows within laboratories and healthcare facilities.
To learn more about this report, Request Free Sample
Within the method segment, Automated Diagnostics is expected to dominate the market throughout the forecast period. The growth of this segment is attributed to the rising adoption of rapid testing technologies that provide faster and more reliable results compared to conventional diagnostic methods. Automated platforms improve efficiency and reduce human error, thereby enhancing patient outcomes.
Conversely, Conventional Diagnostics continues to serve an important role, especially in resource-limited settings where automated solutions may not be readily accessible.
The Bacterial Sepsis segment, encompassing both gram-negative and gram-positive bacterial infections, is anticipated to hold a dominant share in the market. This dominance is largely driven by the increasing prevalence of bacterial infections globally, which constitute the majority of sepsis cases. The high clinical urgency associated with bacterial sepsis fuels demand for effective diagnostic solutions.
Segments such as Fungal Sepsis and Others remain important for addressing less common but clinically significant infections, contributing to a comprehensive diagnostic approach.
Among technology segments, Immunoassays are expected to lead the market owing to their widespread use in rapid sepsis diagnosis. Immunoassay techniques enable quick detection of sepsis biomarkers, supporting early intervention and better prognosis. The growing preference for rapid testing methods further bolsters the immunoassay segment’s position.
Other technologies, including Microbiology, Molecular Diagnostics, and Flow Cytometry, provide critical complementary diagnostic capabilities, enabling pathogen identification and detailed immune profiling.
The Hospitals segment is projected to dominate the end-user market during the forecast period. This is attributed to the critical need for prompt and accurate diagnosis of sepsis in hospital settings, where rapid treatment initiation can significantly improve patient recovery rates. Hospitals serve as the primary point of care for sepsis patients, driving demand for advanced diagnostic tools.
Other end users such as Pathology & Reference Laboratories and Research Laboratories & Academic Institutes contribute to the market by supporting confirmatory testing, research, and development of new diagnostic methods.
Overall, the global sepsis diagnostics market is strongly led by the Instruments segment under product type, driven by regulatory approvals and technological advancements, while other segments complement the diagnostic ecosystem to address the growing clinical demand for rapid and accurate sepsis detection worldwide.
To learn more about this report, Request Free Sample
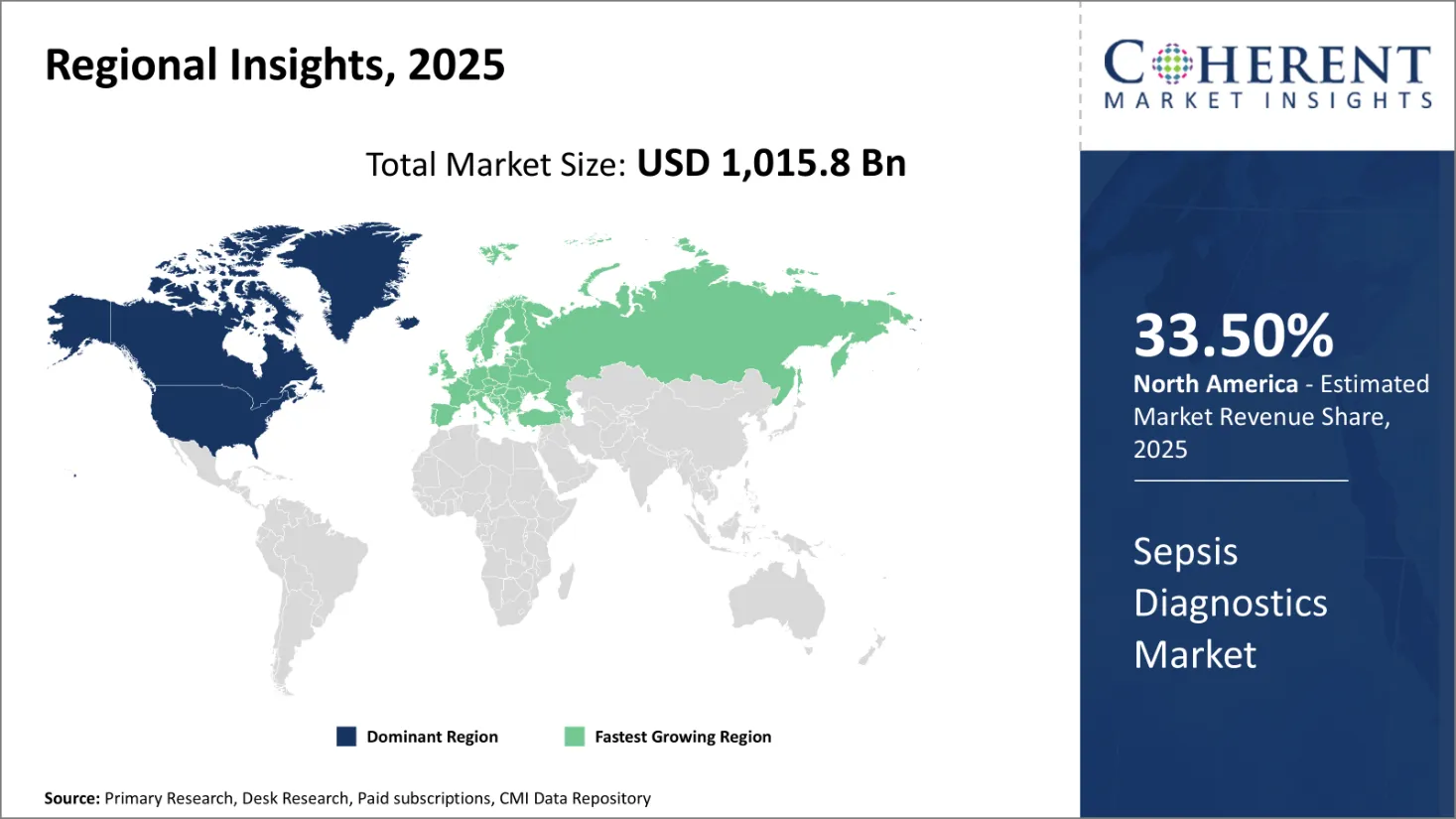
North America is poised to maintain its dominant position in the global sepsis diagnostics market, expected to hold approximately 33.5% of the market share by 2026. The region’s leadership is supported by significant funding opportunities for research and development activities spearheaded by key market players and biotechnology firms.
For example, on June 8, 2021, CARB-X, a U.S.-based biotechnology company, secured funding of up to USD 578,000 to develop novel fiber optic technology aimed at diagnosing sepsis or assessing the risk of sepsis. The project could potentially receive an additional USD 2.1 million contingent upon achieving certain milestones and available funds.
The emphasis on rapid diagnostic testing is especially critical in sepsis care, where every hour of delay in appropriate treatment drastically reduces patient survival rates. North America’s advanced healthcare infrastructure, presence of innovative biotechnology firms, and active government and private sector support continue to drive robust growth in this market.
Europe is witnessing steady growth in the sepsis diagnostics market, supported by its well-established healthcare systems and stringent regulatory frameworks encouraging innovation. Increasing adoption of rapid diagnostic technologies and investments in molecular and immunoassay-based testing contribute to the region’s expansion. Additionally, heightened focus on antimicrobial resistance and infection control measures is encouraging healthcare providers to adopt advanced sepsis diagnostic solutions.
The United States leads the North American market with a robust biotechnology sector and strong governmental support for infectious disease research. The demand for rapid and accurate sepsis diagnostic tools is high across hospitals and reference laboratories, underpinning steady market growth.
China is a key growth driver in the Asia Pacific market, bolstered by expanding healthcare infrastructure, rising incidence of infectious diseases, and increasing adoption of innovative diagnostic technologies. Domestic companies are intensifying R&D efforts to meet growing clinical needs.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1,118.4 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 10.1% | 2033 Value Projection: | USD 2,193.3 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
BioMérieux SA, Thermo Fisher Scientific, Abbott Laboratories, Roche Diagnostics AG, Luminex Corporation, CytoSorbents Corporation, Becton Dickinson Company, Danaher Corporation, Axis-Shield Diagnostics, T2 Biosystems, EKF Diagnostics, Immunexpress Inc., Response Biomedical Corporation, and Bruker Corporation |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The increasing prevalence of infectious diseases is expected to propel the growth of the global sepsis diagnostics market over the forecast period. For instance, on July 19, 2023, according to the World Health Organization (WHO), regional disparities in sepsis incidence and mortality existed; approximately 85% of sepsis cases and sepsis-related deaths worldwide occurred in low- and middle-income countries. Health care-associated infections are one of the most frequent types of adverse events to occur during care delivery and affect hundreds of millions of patients worldwide every year.
Increasing product approvals by regulatory authorities is expected to drive the growth of the global sepsis diagnostics market in the near future.
For instance, on January 18, 2025, Cytovale, a medical diagnostics company, announced that its IntelliSep test has received U.S. Food and Drug Administration (FDA) 510(k) clearance to aid in the early detection of sepsis for the approximately 30 million adult patients with signs and symptoms of infection who present to US emergency departments (ED) each year.
Research institutes in the region are focused on the research and development activities which are expected to drive market growth in the North America region. For instance, on July 13, 2022, researchers at the University of Florida created and are using a diagnostic tool that leverages artificial intelligence to identify a patient’s likelihood of developing sepsis and how severe it will be as soon as 12 hours after the patient’s hospital admission.
The introduction of research and development with advanced technology in the market is expected to drive growth of the global sepsis diagnostics market over the forecast period. For instance, on October 28, 2022, DeepUll, a Spanish molecular diagnostics company, focused on developing molecular test to guide therapy selection for patients with sepsis.
The DeepUll Company announced that it raised USD 14.2 million in Series B financing to fund its efforts to offer a multiplex PCR-based alternative to blood culture, still the go-to technology for diagnosing and treating sepsis patients.
Low adherence to guidelines in sepsis care puts patients at risk and is expected to hinder market growth over the forecast period. For instance, on September 22, 2022, according to bioMérieux, a company in vitro diagnostics with a long-standing commitment to the fight against sepsis, and the UK Sepsis Trust, an internationally renowned charity, released new survey data that was commissioned among 368 doctors across six European countries stated that sepsis is a life-threatening condition in which the body’s response to infection causes organ damage. Every year in Europe, more than 3.4 million people develop sepsis, and 700,000 of them do not survive.
Early recognition and rapid diagnosis are essential for early patient management. Any delay in starting appropriate treatment is associated with increased mortality. A set of evidence-based clinical practices, known as sepsis bundles, have been defined internationally. The routine implementation of these bundles by clinicians can greatly improve outcomes for patients with sepsis.
Key players focusing on developing awareness programs and advanced products with safety and cost-effective treatment would help to drive market growth over the forecast period.
*Definition: Sepsis is a life-threatening medical emergency caused by the body’s overwhelming response to an infection. Without urgent treatment, it can lead to tissue damage, organ failure, and death. Sepsis is the body’s extreme reaction to an infection. When patients have an infection, immune systems work to try to fight it. But sometimes the immune system stops fighting the infection and starts damaging normal tissues and organs, leading to widespread inflammation throughout the body.
Share
Share
Nikhilesh Ravindra Patel is a Senior Consultant with over 8 years of consulting experience. He excels in market estimations, market insights, and identifying trends and opportunities. His deep understanding of the market dynamics and ability to pinpoint growth areas make him an invaluable asset in guiding clients toward informed business decisions. He plays a instrumental role in providing market intelligence, business intelligence, and competitive intelligence services through the reports.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients