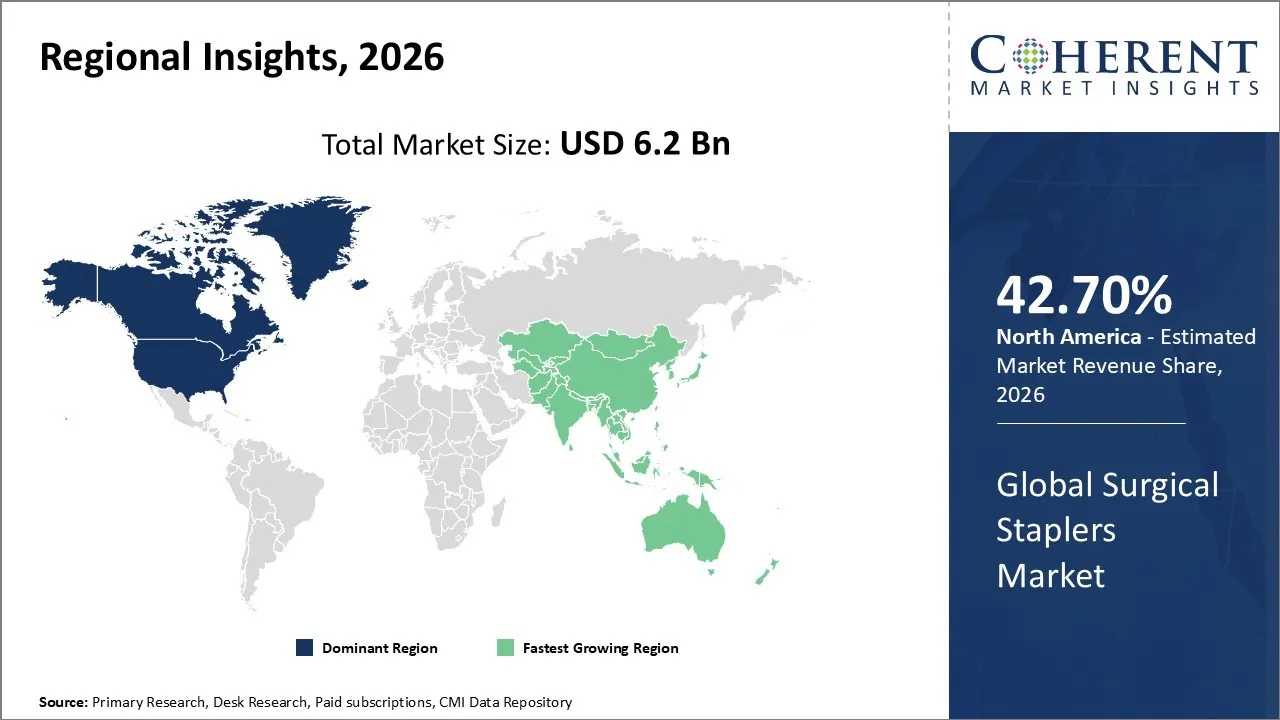
The Surgical Staplers Market is estimated to be valued at USD 6.2 Bn in 2026 and is expected to reach USD 9.55 Bn by 2033, growing at a compound annual growth rate (CAGR) of 6.4% from 2026 to 2033.
The surgical staplers market is advancing significantly with the rising demand for minimally invasive procedures and powered stapling systems in the gastrointestinal, thoracic, and gynecological sectors. The growing surgeon awareness regarding procedural efficiency and a strategic shift away from traditional manual suturing are expected to propel market growth over the forecast period. Surgical Staplers are primarily utilized for rapid tissue approximation and the creation of secure anastomoses. They serve as a high performance alternative to hand-sewn closures, thereby offering superior consistency and reduced operative time.
The industry is entering a transformative period owing to the use of advanced electronic firing technologies, a surge in robot-assisted surgeries, and the integration of smart sensors that provide real-time feedback on tissue thickness. These advancements enhance the precision, safety, and ergonomics of single-use and articulating staplers. As a result, the healthcare providers can replace manual techniques with automated botanical-compatible or titanium-based solutions that assist in catering to the growing global regulatory standards for patient safety and clinical transparency.
|
Current Event |
Description and the Impact |
|
Technological Innovations and Advancements |
|
|
Regional Healthcare Infrastructure Developments
|
|
|
Strategic Industry Movements |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
In terms of product, the linear stapler segment contributes the highest share of 39.50% in 2026 of the market. The growth is owing to its versatility and its effectiveness in open as well as minimally invasive surgeries. These devices are primarily used for tissue resection and transection, delivering consistent straight staple lines that support secure closure and reduce the risk of leaks. The growth of linear staplers is due to their broad applicability in gastrointestinal, thoracic, and bariatric procedures. Furthermore, the development of linear cutter staplers, which can simultaneously staple and divide tissue, has significantly improved surgical efficiency and patient safety. The increasing volume of complex internal surgeries worldwide highlights the precision and reliability of linear staplers, which are favored by surgeons seeking to reduce operative time and postoperative complications.
In terms of application, the abdominal surgery segment contributes the highest share of 41.80% in 2026 of the market. The growth of this sector can be mainly attributed to the high prevalence of gastrointestinal disorders and colorectal cancers as well as the rising demand for bariatric and weight loss surgeries. The surgical staplers in abdominal procedures is owing to their capability to provide faster and more reliable outcomes compared to hand suturing, especially when creating complex bowel anastomoses. The increasing use of laparoscopic abdominal techniques has also boosted growth, resulting in higher utilization of specialized staplers designed for minimally invasive access. The implementation of enhanced recovery protocols aims to reduce hospital stays and minimize the risk of infections. This strategy is promoting the increased use of staplers in high-volume abdominal surgical settings around the globe.
In terms of mechanism, the manual surgical stapler segment contributes the highest share of 61.20% in 2026 of the market. The growth is due to its cost-effectiveness and widespread availability, especially in healthcare systems with budget constraints and in emerging economies. The preference surgeons show for manual staplers is owing to the tactile feedback these devices provide, which enables real-time assessment of tissue thickness and procedural control. These devices play a pivotal role in routine surgical procedures owing to their simple design, absence of electronic components, and well-established safety profile. The manual staplers continue to play a central and indispensable role in general surgery.
To learn more about this report, Request Free Sample
North America has remained the dominant region with 42.70% in 2026 of the global surgical staplers market over the forecast period. The growth is owing to the substantial public health investments and a strong healthcare infrastructure that enables prompt diagnosis. The implementation of early screening programs like Early Hearing Detection and Intervention (EHDI) is also constituting to the growth of the market in the region. The technological breakthroughs serves as a vital catalyst for growth, particularly through the rising adoption of minimally invasive surgical (MIS) techniques, robotic-assisted navigation systems, and AI-powered diagnostic tools that improve precision and reduce recovery times.
For instance, in June 2025, Johnson & Johnson MedTech announced that the ETHICON™ 4000 Stapler will soon be available in the US. This cutting-edge surgical stapler is made to handle tissue complexity and provide outstanding staple line integrity.
The Asia Pacific region is the fastest growing market for surgical staplers. The growth is owing to an increase in minimally invasive surgeries (MIS) and the increasing aging population in countries like Japan, China, and South Korea. India is experiencing growth owing to the rapid expansion of private multi-specialty hospitals and a thriving medical tourism sector. This sector attracts international patients in search of cost-effective bariatric and cardiac procedures. The competitive landscape encompasses both established global firms as well as emerging local businesses. The top companies like Medtronic, Johnson & Johnson (Ethicon), and Intuitive Surgical are incorporating advanced technologies like real-time tissue-sensing algorithms and powered firing mechanisms to improve procedural accuracy.
For instance, in January, 2025, Genesis Medtech has announced that the iReach Omnia has obtained clearance for market release in China from the National Medical Products Administration (NMPA). This is the first powered stapler that features the ability to articulate at a 90° angle.
The U.S. surgical staplers market is a highly mature sector defined by the widespread use of advanced medical technologies and a robust healthcare infrastructure. The market is currently undergoing a major transition toward powered and electronic stapling systems that integrate smart sensors to monitor tissue thickness during complex procedures. This surge is primarily due to the high prevalence of obesity and chronic diseases, which has resulted in a substantial volume of bariatric and thoracic surgeries. In addition, the rapid expansion of robot-assisted surgery across American hospitals necessitates specialized articulating staplers designed for precision. The stringent safety regulations and a strong emphasis on reducing operating room time are encouraging the use of high-quality, disposable instruments.
For instance, in June 2022, Ethicon, a division of Johnson & Johnson MedTech, has announced the availability of the ECHELON 3000 Stapler in the U.S. Surgeons can tailor simple, one-handed powered articulation to meet the specific needs of each patient using this digitally enabled tool.
The Indian surgical staplers market is characterized by rapid expansion and a diversifying landscape that balances cost-sensitivity with a demand for modern technology. Although traditional manual staplers remain a staple in many public healthcare facilities due to their affordability, there is an accelerating trend toward minimally invasive techniques in urban private hospitals. A growing middle class, an increase in elective surgeries, and India's status as a global hub for medical tourism are the main factors driving the market. The government initiatives aimed at expanding surgical coverage have also increased the penetration of specialized wound closure tools in secondary cities.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 6.2 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 6.4% | 2033 Value Projection: | USD 9.55 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Grena Ltd., 3M Company, Smith & Nephew, Purple Surgical Inc., Meril Life Science Pvt. Ltd., Medtronic PLC, Intuitive Surgical Inc., B. Braun Melsungen AG, CONMED Corporation, Frankenman International Limited, and Ethicon Inc. (Johnson & Johnson), among others |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The global shift toward minimally invasive surgery (MIS) has become the primary catalyst for the surgical staplers market, fundamentally transforming how surgeons manage wound closure and tissue handling. MIS techniques such as laparoscopy and thoracoscopy rely on small entry points that necessitate highly specialized instruments. In these limited spaces, traditional hand-suturing often proves to be technically challenging and time-consuming. As a result, surgical staplers have surfaced as the preferred option, providing a mechanical solution that ensures rapid, consistent, and secure closure of internal tissues and vessels through narrow trocars.
An increasing amount of clinical evidence supports the use of these procedures because they lead to better patient outcomes, such as less pain after surgery, shorter hospital stays, and faster returns to normal activities. As healthcare systems worldwide shift toward value-based care, the efficiency that staplers offer becomes essential. They substantially reduce operating room time, consequently lowering the overall cost per procedure. The growth of robotic-assisted surgery, exemplified by systems such as Intuitive Surgical's da Vinci platform, has integrated advanced stapling technology into robotic arms. These sophisticated staplers utilize powered firing systems and tissue-sensing technology to ensure that staples are applied with the appropriate amount of pressure.
The global Surgical Staplers Market is experiencing sustained growth driven by increasing surgical procedure volumes, particularly in gastrointestinal, bariatric, and thoracic domains, where staplers improve efficiency and consistency compared with traditional suturing. Disposable staplers account for a significant share of usage, often cited at 60–65%, due to sterility considerations and ease of operation, while powered staplers are witnessing faster adoption owing to enhanced precision and control. Hospitals remain the largest end-user segment, representing approximately 70–75% of demand, as a result of high procedural volumes and complex surgeries.
Regionally, North America is the leading market, supported by advanced healthcare infrastructure and high adoption of minimally invasive techniques. In contrast, the Asia-Pacific region is the fastest-growing, fueled by expanding hospital networks, rising surgical procedure volumes, and increasing healthcare accessibility.
Technological innovations are shaping market dynamics, including smart stapling systems providing real-time feedback, ergonomic designs, and integration with robotic surgical platforms. Outpatient and ambulatory surgical centers are also increasingly adopting staplers to enhance procedural efficiency.
Overall, market expansion is underpinned by strong clinical demand, continuous technological advancements, and the growing global emphasis on surgical precision and operational efficiency, positioning surgical staplers as indispensable tools in modern surgical practice.
Definition: The Surgical Staplers Market is a global medical technology sector focused on the design and distribution of mechanical instruments that use specialized fasteners to approximate tissue or close wounds. These devices are favored for traditional suturing owing to their ability to provide rapid, uniform, and secure seals. This helps in substantially reducing operative time and the risk of infection. The industry is shifting toward powered systems that incorporate automated firing and real-time tissue sensing. These features contribute to better surgeon ergonomics and reliable staple formation. In addition, the expansion of minimally invasive and robotic-assisted surgical approaches is driving demand for slim, articulating staplers designed to access and maneuver within constrained anatomical environments. This landscape underscores a structural preference for disposable, single-use formats, thereby aligning with broader efforts to improve patient safety and reduce sterilization-related labor and infrastructure costs.
Share
Share
Manisha Vibhute is a consultant with over 5 years of experience in market research and consulting. With a strong understanding of market dynamics, Manisha assists clients in developing effective market access strategies. She helps medical device companies navigate pricing, reimbursement, and regulatory pathways to ensure successful product launches.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients