Elastomeric infusion pumps are also called as balloon pumps, which are generally used to administer liquid drugs such as local anesthetics, analgesic, cytostatic, and antibiotics. Elastomeric pumps do not require any electricity, when these pumps are placed on the patient the flow rate of the pumps is typically increased or decreased, which means the elastomeric pumps are manually operated pumps and also the not affected by gravity . The elastomeric pumps are maintenance free due to no use of electricity and are single use disposable.
The U.S. elastomeric infusion pumps market is estimated to be valued at US$ 258.3 million in 2021 and is expected to exhibit a CAGR of 5.7 % during the forecast period (2022-2028).
Figure 1. U.S. Elastomeric Infusion Pumps Market Value (US$ Mn), 2017-2028
To learn more about this report, Request Free Sample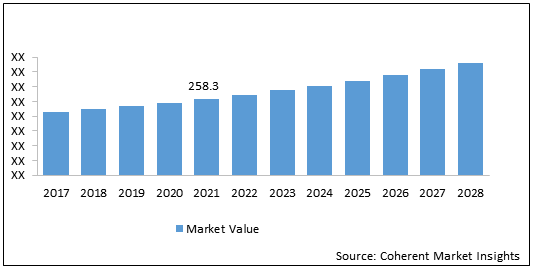
The increasing incidence of chronic diseases is expected to drive the market growth over the forecast period. According to the Centers for Disease Control and Prevention Report published on September 2020, heart disease is the leading cause of death for men, women, and people of most racial and ethnic groups in the U.S., and one person dies every 36 seconds in the U.S. from cardiovascular disease.
Moreover, the increasing R&D for novel applications of elastomeric infusion pumps is expected to drive the market growth over the forecast period. For instance, in September 2018, researchers from University Hospital of Lausanne, Switzerland, assessed the use of two sub-muscular catheters connected to two elastomeric pumps in post-operative continuous wound.
U.S. Elastomeric Infusion Pumps Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2020 | Market Size in 2021: | US$ 258.3 Mn |
| Historical Data for: | 2017 to 2020 | Forecast Period: | 2020 to 2027 |
| Forecast Period 2022 to 2028 CAGR: | 5.7% | 2028 Value Projection: | US$ 380.7 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
B. Braun Melsungen AG, Baxter International Inc., Werfen, Ambu A/S, Avanos Medical Inc., NIPRO, and Progressive Medical, Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
U.S. Elastomeric Infusion Pumps Market – Impact of Coronavirus (COVID-19)
Following the outbreak of COVID-19 in December 2019, the disease has spread to over 100 countries across the globe and the World Health Organization declared it as a public health emergency. According to the World Health Organization’s report, manifestation of coronavirus (COVID-19) has resulted in over 162 million infected individuals worldwide as of May 17, 2021.
The coronavirus (COVID-19) pandemic and resulting lockdown in various countries across the globe has impacted the financial status of major companies in urea cycle disorders product market. The private healthcare sector is one of the sectors, which has been majorly impacted by the COVID-19 pandemic. The pandemic has negatively impacted the U.S. elastomeric infusion pumps market in various aspects such as research and development, production, and supply of medicines. The pandemic has also affected the growth of pharmaceutical businesses of various companies across the globe due to lockdowns implemented by governments of several countries.
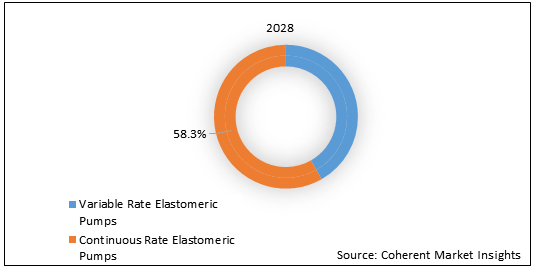
Figure 2. U.S. Elastomeric Infusion Pumps Market Share (%), By Product Type, 2028
To learn more about this report, Request Free Sample
Moreover, stringent regulations regarding the use of plastics in elastomeric infusion pumps is expected to hamper the market growth. For instance, the European Commission and the U.S. FDA published safety assessments of medical devices containing Di (2-ethylhexyl) phthalate (DEHP) and plasticized polyvinylchloride (PVC) in certain parts of elastomeric infusion pumps.
Therefore, manufacturers need to focus on proper safety labelling of products to avoid severe side effects, especially in neonatal patient population.
Furthermore, the cost of elastomeric infusion pumps keep fluctuating depending on the type of the pump.
For instance, the cost of pumps administering antibiotics is significantly low, whereas the cost of pumps administering pain management drugs is relatively high and ranges between US$ 160 and US$ 450. Such factors is expected to hinder growth of the U.S. elastomeric infusion pumps market over the forecast period.
Key Players
Major players operating in the U.S. elastomeric infusion pumps market include B. Braun Melsungen AG, Baxter International Inc., Werfen, Ambu A/S, Avanos Medical Inc., NIPRO and Progressive Medical, Inc.
Share
Share
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients