The global intravenous access devices market size is expected to reach approximately USD 62,350 Mn in 2026 and is projected to grow at a compound annual growth rate (CAGR) of 6.6% during the forecast period (2026-2033), reaching USD 97,750 Mn by 2033. This is growth attributable to rising prevalence of chronic diseases requiring intravenous therapy and advancements in minimally invasive medical devices.
The global intravenous access devices market is expected to grow steadily during the forecast period. This is primarily due to rising prevalence of chronic diseases, increasing number of surgical procedures, and growing demand for minimally invasive therapeutic interventions. In addition, expansion of hospital infrastructure, advancements in catheter and infusion technologies, and heightened awareness regarding infection prevention and patient safety are supporting market expansion.
Intravenous access devices (IVADs) are small, flexible tubes placed in veins to provide long-term access to the bloodstream for people who require frequent or continuous medication, fluid, or blood administration. Some IVADs, such as totally implantable venous ports, are implanted under the skin and connect to central veins in the chest or neck.
IVADs, which include catheters and ports, also make repeated access to the bloodstream easier and less painful, reducing the need for multiple needle sticks. Rising adoption of these medical devices in hospitals, clinics, home care, and other healthcare facilities is expected to provide a strong impetus for the growth of intravenous access devices market during the forecast period.
Shift towards minimally invasive and patient-centric solutions is supporting expansion of the intravenous access devices market. There is rising demand for minimally invasive, patient-friendly devices that increase comfort, reduce complication risks, and support outpatient care. Similarly, growth in home infusion therapy is fueling demand for intravenous access devices. As healthcare shifts toward home-based treatment models, demand grows for user-friendly IV access solutions suitable for non-clinical settings.
|
Current Event |
Description and its Impact |
|
Global Aging Population and Chronic Disease Surge |
|
|
Technological Advancements in Smart Medical Devices |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
IV catheters are expected to remain the top-selling product type in the market, accounting for a revenue share of 50% in 2026. This is mostly due to increasing use of intravenous catheters in hospitals and clinics for short-term and long-term intravenous therapy, ease of insertion, and cost-effectiveness compared to other intravenous access devices.
Rising prevalence of chronic diseases, increasing surgical procedures, and growing demand for minimally invasive treatments are driving the continuous adoption of IV catheters across various healthcare settings. Innovations such as safety-engineered catheters and advanced materials that reduce infection risks further reinforce their dominant position in the global market.
According to Coherent Market Insights, hospitals and clinics are anticipated to remain the leading end users of intravenous access devices during the forecast period, holding a market share of 70% in 2026. This is attributable to high volume of inpatient and outpatient procedures requiring intravenous therapy, as well as the increasing prevalence of chronic and acute diseases.
Rising demand for advanced IV technologies that improve patient care and reduce complications is driving intravenous adoption in hospitals and clinics. In addition, hospitals and clinics usually have the infrastructure, trained staff, and resources to support widespread use of devices like IV catheters, infusion pumps, and needles, thereby boosting growth of the target segment in the market.
To learn more about this report, Request Free Sample
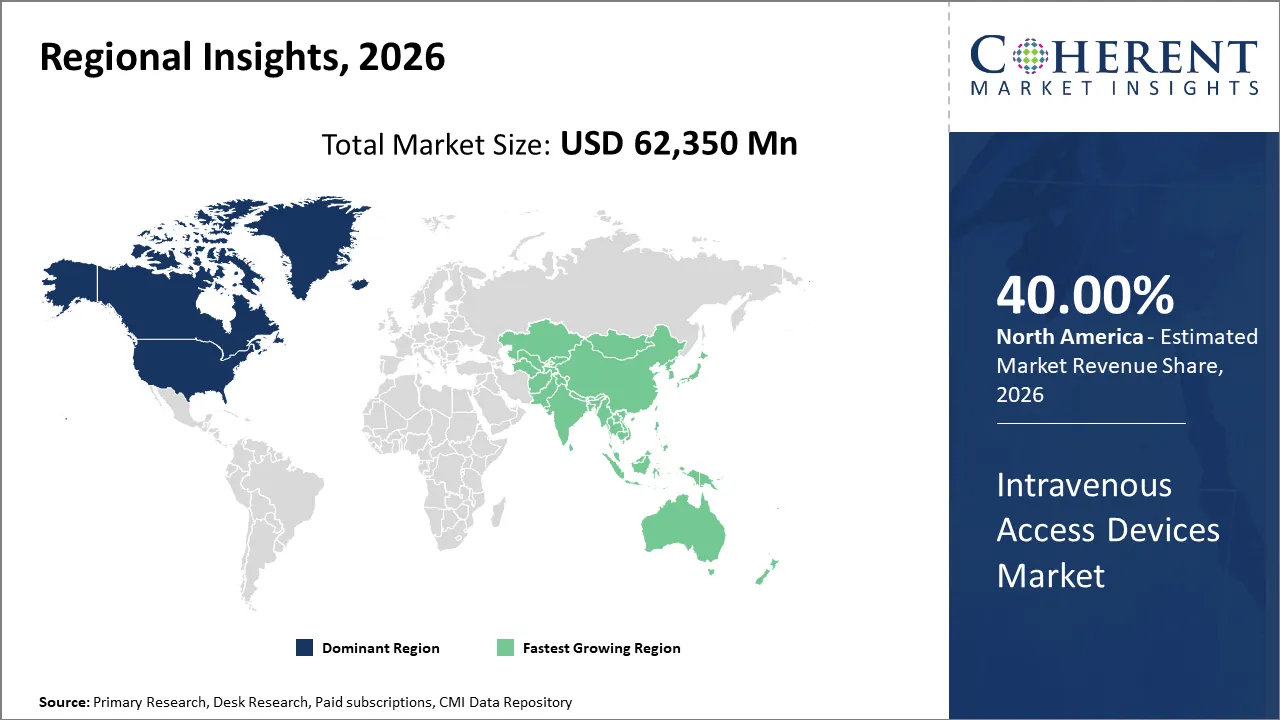
North America is anticipated to retain its dominance over the global intravenous access devices market, capturing a share of 40% in 2026. This is mostly due to high adoption of advanced IV technologies such as smart infusion pumps, safety IV catheters, and automated monitoring systems. Hospitals are increasingly focusing on reducing IV-related complications, improving patient safety, and streamlining workflow efficiency, which drives the demand for reliable intravenous access devices.
Strong presence of some of the top medical device manufacturers, including Becton, Dickinson and Company, Teleflex, Inc., C.R. Bard, Inc., Smiths Medical, Inc., and Baxter International Inc. in the U.S. is also fostering market growth. Government initiatives supporting hospital modernization and the widespread use of outpatient surgical centers further boost market expansion. In addition, North American hospitals are increasingly implementing stringent patient safety protocols, which encourage the adoption of high-quality IV devices.
Asia Pacific is emerging as the fastest‑growing regional market for intravenous and vascular access devices in the world. This rapid growth is mainly driven by increasing cases of chronic diseases (such as cancer, diabetes, cardiovascular disorders), a growing elderly population, and expanding healthcare infrastructure and hospital care needs across countries like China, India, and Japan. The region is likely to account for 20% of global intravenous access device sales in 2026.
Asia Pacific is experiencing a rise in chronic diseases like diabetes and cardiovascular disorders, which often need frequent IV treatments, driving demand. For example, the International Diabetes Federation estimates that adults with diabetes in China will reach 168.3 million by 2050. This high diabetes burden is expected to increase sales of intravenous access devices during the forecast period.
Rising demand for long‑term intravenous therapies, including chemotherapy, dialysis, and critical care treatments, is significantly increasing the use of vascular access and IV devices in the region. Similarly, rising government spending, expanding hospital networks, and adoption of modern surgical and perioperative care practices are creating strong demand for IV catheters, infusion pumps, and related devices in Asia Pacific.
The U.S. is expected to remain the leading market for intravenous access devices during the forecast period. This is mostly due to advanced healthcare infrastructure, high patient volumes, and widespread adoption of safety-focused IV technologies, including smart infusion pumps and safety catheters. Hospitals are increasingly implementing protocols to reduce IV-related complications and improve workflow efficiency, which is driving demand for reliable intravenous access devices.
Rising emphasis on patient safety and technological adoption is providing a strong impetus for the growth of intravenous access devices market in the U.S. Hospitals and outpatient surgical centers are investing in automated IV monitoring systems and advanced catheters to improve treatment outcomes.
The intravenous access devices market in China is poised to exhibit robust growth during the forecast period. This can be attributed to rapid expansion of modern hospitals, rising surgical volumes, and increased healthcare spending. The government’s Healthy China 2030 initiative is supporting hospital modernization and the adoption of advanced medical devices.
Rise of minimally invasive and robotic-assisted surgeries in tier-1 and tier-2 cities is also playing a key role in boosting intravenous access devices market value in China. These procedures require precise intravenous access and monitoring, boosting demand for smart infusion pumps, safety catheters, and related devices across hospitals and surgical centers.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 62,350 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 6.6% | 2033 Value Projection: | USD 97,750 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Becton, Dickinson And Company, Teleflex, Inc., C.R. Bard, Inc., Fresenius Medical Care, Smiths Medical, Inc., Hospira, Inc., Covidien Plc, Fresenius Medical Care AG & Co., and Baxter International Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Ambulatory care centers and home healthcare services are expanding significantly, especially across developing regions like Asia Pacific. This, in turn, is driving demand for portable, easy to use IV access devices that support treatments outside hospitals. Thus, rising adoption of home care services is expected to create lucrative growth opportunities for the intravenous access devices during the forecast period.
Innovation is at the forefront of the intravenous (IV) access devices market. Advancements such as smart monitoring systems, needle-free access technologies, and safer catheter materials enhance patient safety, improve dosing accuracy, and simplify clinical workflows, driving greater adoption in healthcare settings.
Leading manufacturers of intravenous access devices are introducing novel solutions like ultrasound-guided IV catheters, antimicrobial-impregnated catheters, and wearable IV infusion systems. These innovations not only reduce the risk of infection and improve patient comfort but also enable real-time monitoring of fluid delivery, making IV therapy more efficient and reliable.
Share
Share
Manisha Vibhute is a consultant with over 5 years of experience in market research and consulting. With a strong understanding of market dynamics, Manisha assists clients in developing effective market access strategies. She helps medical device companies navigate pricing, reimbursement, and regulatory pathways to ensure successful product launches.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients