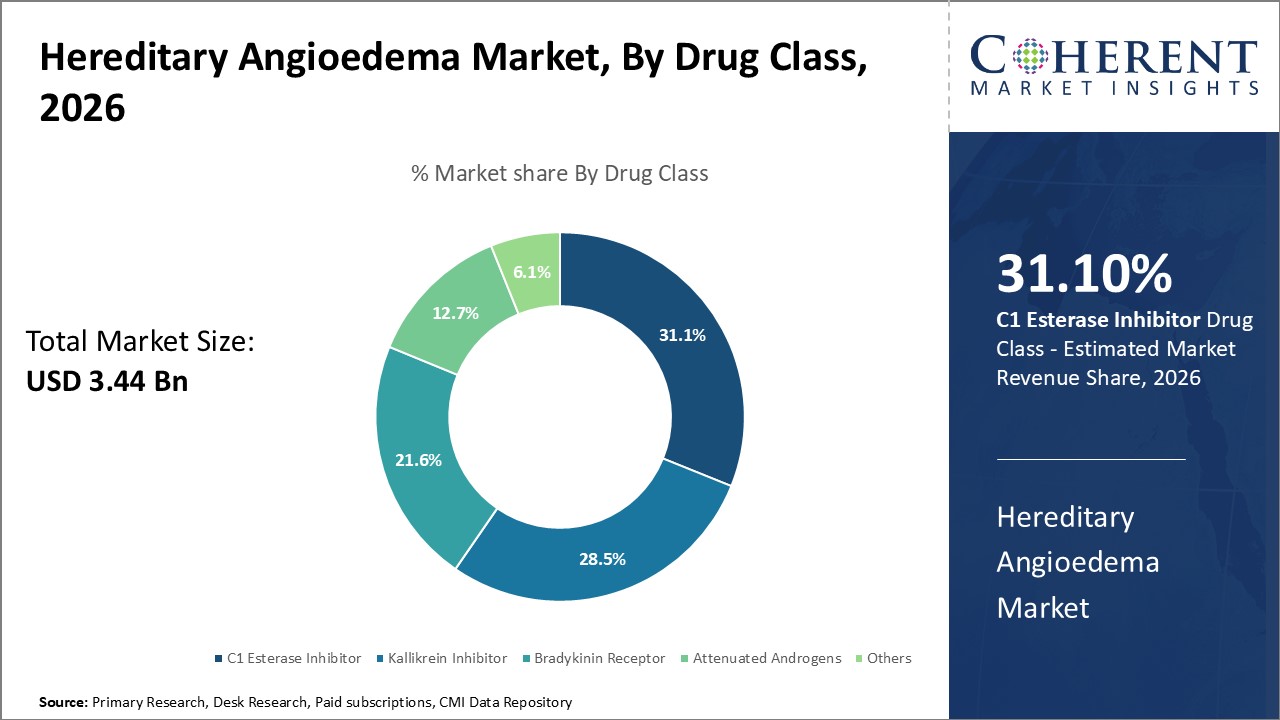
Hereditary Angioedema Market size is estimated to be valued at USD 3.44 Bn in 2026 and is expected to reach USD 6.61 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 9.8% from 2026 to 2033.
To learn more about this report, Request Free Sample
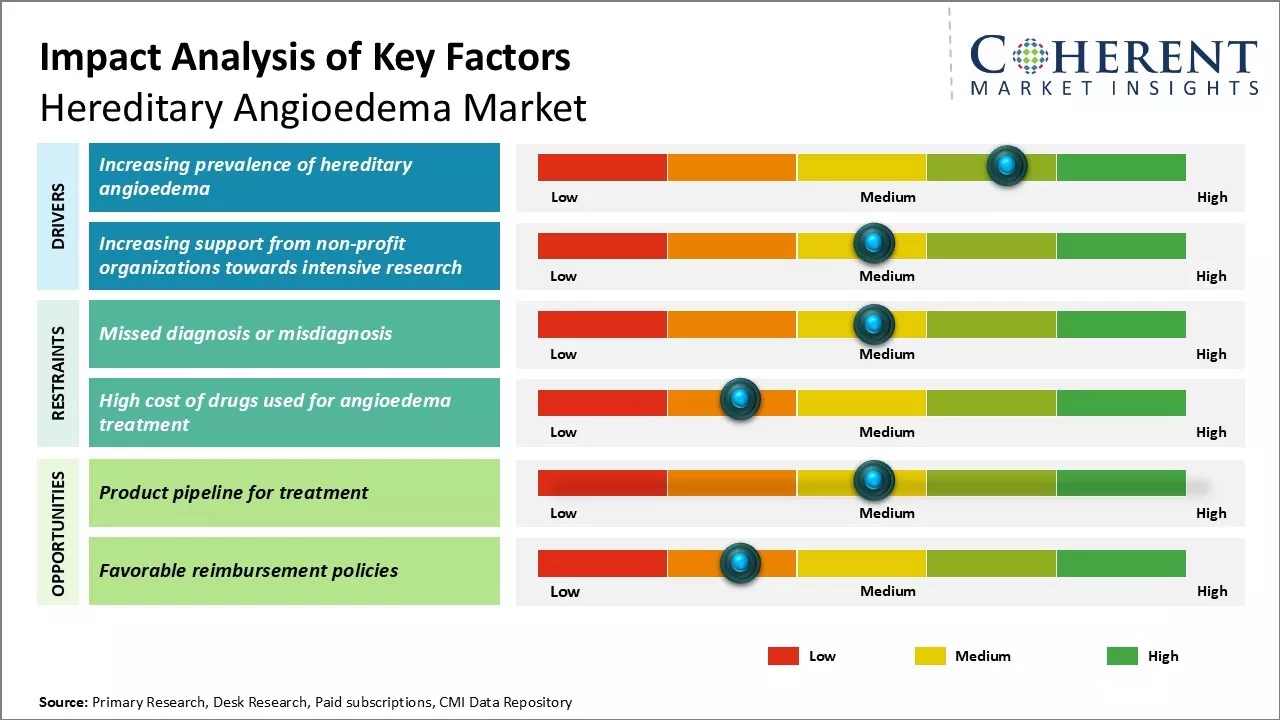
Hereditary Angioedema Market Growth expansion is mostly caused by increasing understanding of the condition as well as the introduction of new treatment alternatives. Newer diagnostic tools have resulted in more patients being accurately and earlier diagnosed, which increases the patient population and the need for therapy. Availability of oral dosage forms allows for easy self-administration at home, which is more convenient than traditional injectables. On the other hand, the market faces some challenges concerning the overall high treatment costs that are often associated with these medications which limits access for a number of patients.
|
Event |
Description and Impact |
|
increasing prevalence of Hereditary Angioedema (HAE) |
|
|
Advancements in HAE treatment options |
|
|
Collaborations and partnerships in the HAE space |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
In terms of type, hereditary angioedema type I segment is estimated to contribute the highest Hereditary Angioedema Market Demand share of 68.8% in 2026, due to increased awareness about the condition. Rising diagnosis rates and awareness initiatives have encouraged patients with mild symptoms to seek treatment. Advancements in targeted therapy have also improved the management of attacks and adherence to treatment protocols.
To learn more about this report, Request Free Sample
In terms of Drug Class, the C1 esterase inhibitor segment will lead the Hereditary Angioedema Market Forecast share with 31.1% in 2026 due to its well-established efficacy and safety profile. This class features the first drug approved by the U.S. FDA for the treatment of acute attacks of HAE with self-administered enhancement patient options. The ability of patients to reconstitute and act within 30 minutes empowers them to cope with initial symptom control, further cementing C1 esterase inhibitors' commanding market position.
In terms of Route of Administration, subcutaneous route of administration will dominate the Hereditary Angioedema Market Outlook by 31.1% share in 2026 due to high patient mobility. The use of self-administered subcutaneous injections has been on the rise among HAE patients, a trend which spares the need for hospital access, relevant to the treatment of mild-to-moderate attacks that happen outside medical facilities.
To learn more about this report, Request Free Sample
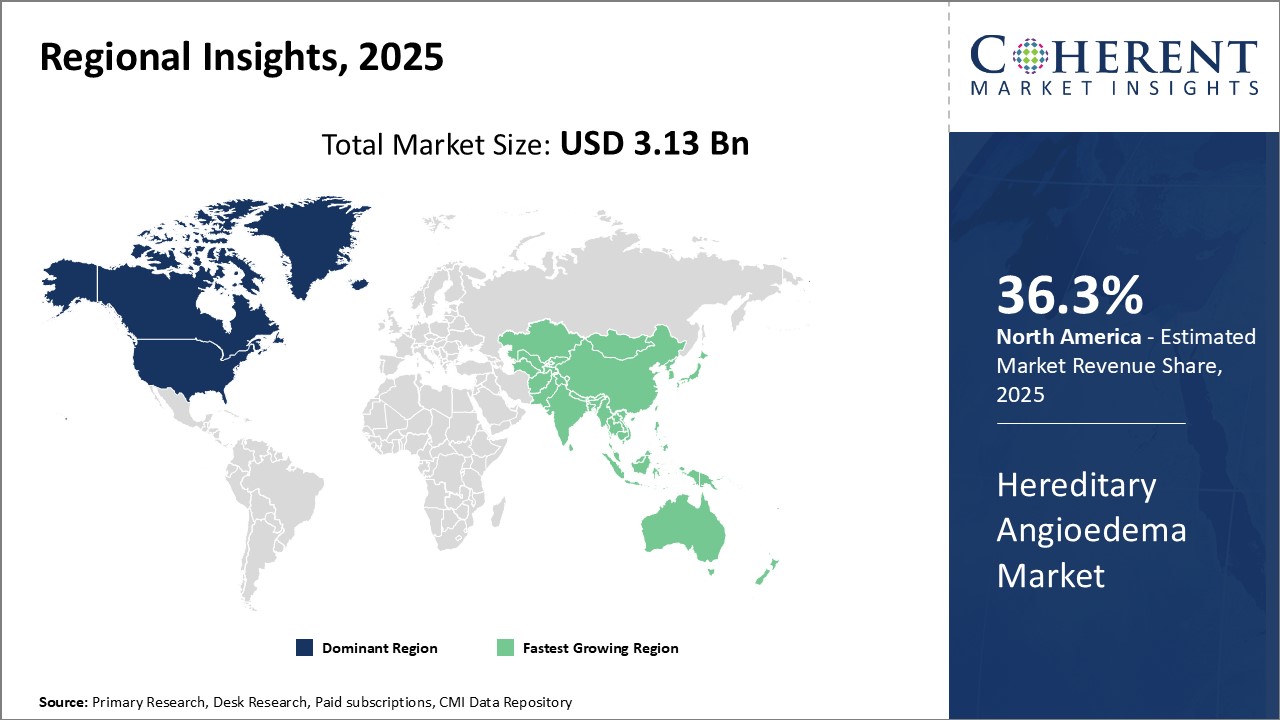
North America is predicted to have the largest Hereditary Angioedema Market Value controlling an approximate share of 36.3% in 2026. This is attributed to the count of major pharma players in the region, favorable healthcare reimbursement policies, and heightened patient awareness.
The Asia Pacific region is expected to grow at the fastest rate capturing an approximate market share of 22.9% in 2026. This is due to the evolving healthcare system, better access to medical services, and expansion efforts by other major players in the emerging markets.
Innovative therapies are constantly being introduced in the United States, which is greatly aiding the growth of the Hereditary Angioedema Market Trends. Companies are now focusing precision medicine on individual patients’ needs by investing more in clinical trials to develop treatment regimens through targeted therapies. This shift is important as it leads to the development of next-generation therapies such as oral and injectable subcutaneous medications that are more convenient and easier to follow than existing therapies.
The growth of the German hereditary angioedema sector is facilitated by encouraging national policies and support directed toward the management of rare diseases. The German government is bolstering healthcare for rare diseases through its National Action Plan which aims at improving the overall strategy for diagnosis, treatment, and patient management. The partnership of healthcare providers and pharmaceutical companies actively supports the R&D initiatives to develop new therapies for wider use.
Focus on Japan's clinical practices regarding HAE has driven marked growth for the region. The increased recognition of HAE among professionals issyndicated by focused marketing promoting the diseases. Pharmaceutical firms are increasingly participating in the campaigns to educate doctors about the signs, identifying methods, and treatment options available for HAE. There is notable development from the market leaders directed at new medicines focused on HAE patients’ needs in the Japan hereditary angioedema market.
The expansion of the China hereditary angioedema market is attributed to the growth of investment in the health systems of the country. Medical professionals have been given improved qualifications and extensive access to modern healthcare devices. The reform aims to ensure the entire population can receive medical assistance while solving the issues of healthcare accessibility and quality between urban and rural regions. This goal comes under the wider initiative termed ‘Healthy China 2030’ which aims to provide greater disease prevention.
To learn more about this report, Request Free Sample
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 3.44 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 9.8% | 2033 Value Projection: | USD 6.61 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
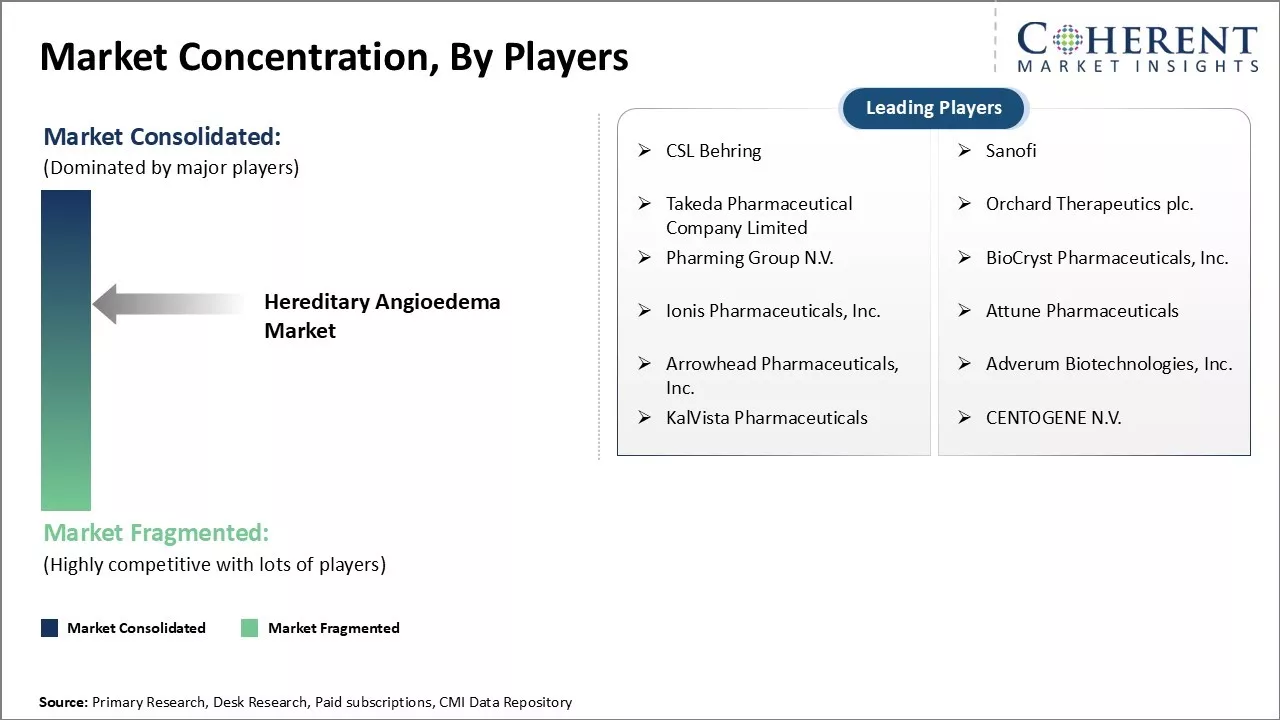
| Companies covered: |
CSL Behring, Sanofi, Takeda Pharmaceutical Company Limited, Orchard Therapeutics plc., Pharming Group N.V., BioCryst Pharmaceuticals, Inc., Ionis Pharmaceuticals, Inc., Attune Pharmaceuticals, Arrowhead Pharmaceuticals, Inc., Adverum Biotechnologies, Inc., KalVista Pharmaceuticals, and CENTOGENE N.V. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The rising prevalence of HAE might cause an increase in economic investment. A systematic review titled “Epidemiology, Economic, and Humanistic Burden of Hereditary Angioedema: A Systematic review” published in 2024 in the Orphanet Journal of Rare Diseases examined more than 65 studies with over 10,000 participants and estimated that the global prevalence of hereditary angioedema (HAE) is between 0.13 to 1.6 cases per 100,000 people in a region. For instance, Austria reported a prevalence of 1.6 per 100,000 while South Korea reported 0.13 per 100,000. Black patients in the US were reported to have a prevalence of 1.64 per 100,000 which is higher than white patients estimated at 1.47 per 100,000
Market opportunity may be restricted by a lack of public understanding about the condition. For example, “What to Know about Hereditary Angioedema (HAE)” is an article published on Health.com in 2025 that discusses the difficulties associated with identifying HAE. Due to its rarity and the shared symptoms of many other diseases, HAE is often misdiagnosed which contributes to further diagnostic delays. The article mentions that patients will likely receive the right diagnosis after a decade or more of having the symptoms which is horrendous to think about. This only
The developed HAE treatment products can easily be exploited by market competitors through their enhanced effectiveness and broadened range of application. Treatment adherence is improved with the innovation of oral medications and subcutaneous injections, along with next-generation prophylactic and on-demand therapies. The incorporation of long-acting C1 inhibitors and Kallikrein inhibitors are proving to be transformative with self-administered and variable dosing which will significantly enhance the life of the patients suffering from HAE. For example, monoclonal antibodies like garadacimab which targets factor XIIa have been promising for long-term prophylactic treatment. A 2024 study supports the notion of subcutaneous monthly injection due to the enduring safety profile of garadacimab during HAE attacks and overall favorable safety.
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients