The Plasma Protease C1-inhibitor Treatment market is anticipated to grow at a CAGR of 9.1% with USD 4.38 Bn in 2026 and is expected to reach USD 8.06 Bn in 2033. The increasing focus of biopharmaceutical companies on development of effective plasma protease C1-inhibitor treatment drugs/therapy treating various health conditions such as HAE, diabetic macular edema (DME) (GlobalData estimates that in 2024, approximately 81% of diagnosed prevalent cases of DME in diabetes were center involving or ci-DME), and others, is expected to drive growth of the plasma protease C1-inhibitor treatment market.
C1-inhibitors is projected to account for the largest share of drug type in 2026, representing approximately 65% of the total volume. C1-inhibitors are extensively used because they work well, and are approved by regulators, as well as being recommended in many HAE treatment guidelines. In 2025, about 1.22 people per 100,000 had HAE, with lower numbers in Asia and Africa compared to Europe and North America.
C1-inhibitors are the most used as they directly replace the missing or faulty C1-esterase inhibitor protein in people with HAE.
This therapeutic approach addresses the root cause of the complement as well as contact system dysregulation, making it the gold standard treatment recommended by leading medical organizations including the World Allergy Organization (WAO), the European Medicines Agency (EMA), etc.
To learn more about this report, Request Free Sample
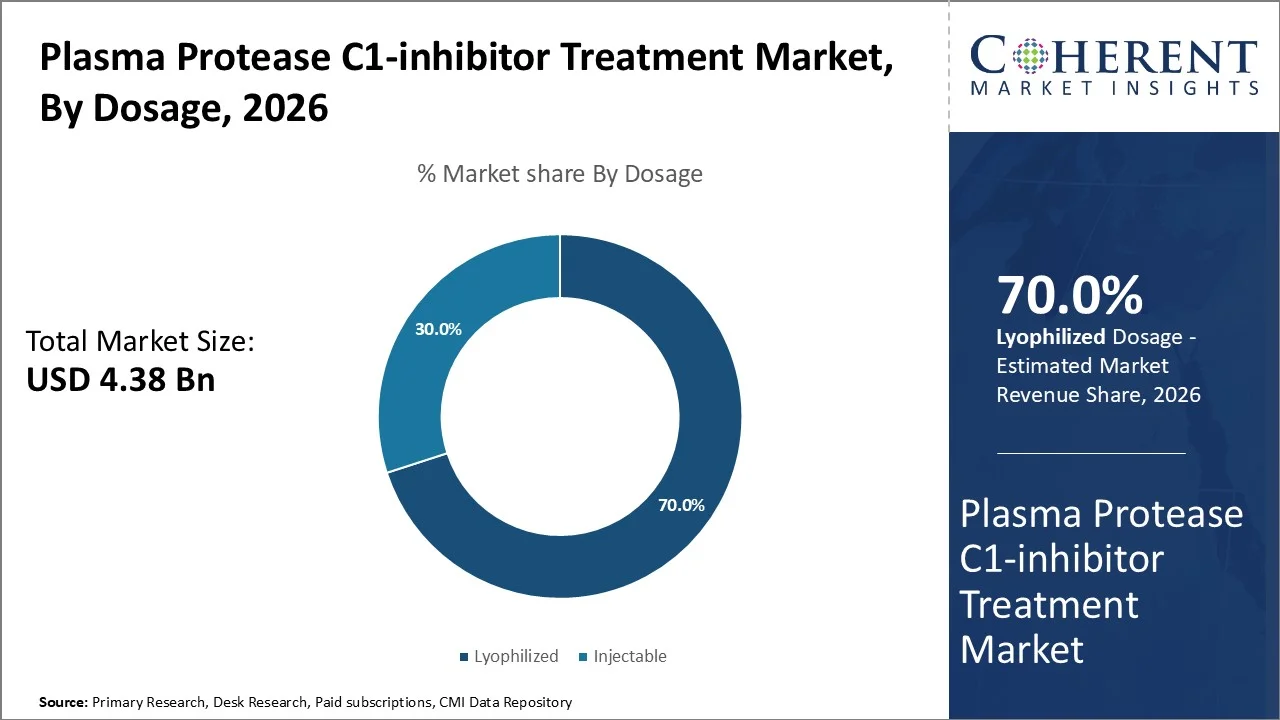
Based on dosage, lyophilized dominates the market, accounting for a significant 70% share in 2026, owing to its superior stability characteristics, extended shelf life, as well as established clinical protocols that have been improved over decades of therapeutic application. The lyophilized formulation of C1-inhibitor reflects a freeze-dried powder that holds the protein's structural integrity as well as biological activity for a longer periods compared with liquid formulations.
According to a study conducted by the National Library of Medicine in 2025, the number of FDA approvals for lyophilized injectables has surged by over 300% since 2000s. Most of the lyophilized medicines are generic small molecule drugs (70% in 2022).
Similarly, over 70% (14 out of 19) of antibiotics on the Essential Medicines list are supplied as lyophilized sterile powders for injection.
C1-inhibitor products need to stay stable because the protein is fragile and can break down in liquid form. The World Health Organization recommends keeping the protein stable during storage and shipping, which is why freeze-dried (lyophilized) forms are usually used around the world.
Based on distribution channel, hospital pharmacies dominates the market, accounting for a significant 60% share in 2026, their dominance reflects the specialized infrastructure and clinical expertise required for administering plasma-derived therapies, particularly for hereditary angioedema patients.
The market’s expansion is supported by strong plasma supply networks. For instance, BioLife Plasma Services, part of Takeda, operates over 260 advanced plasma collection facilities across the U.S. and Europe, making a stable, high-quality plasma supply. This reliable access to plasma makes sure manufacturers to scale production of C1-inhibitor therapies.
Hospital pharmacies serve as the primary distribution hub for C1-inhibitor treatments due to the critical nature of hereditary angioedema (HAE) as well as acquired C1-inhibitor deficiency conditions that usually require instant medical treatment along with specialized storage conditions.
The dominance of hospital pharmacies comes from their ability to maintain the cold chain storage requirements essential for plasma protease C1-inhibitor products, which majorly need refrigeration between 2-8°C to maintain their therapeutic efficacy.
According to the World Federation of Hemophilia, specialized treatment centers and hospital-based facilities handle approximately 70% of plasma-derived therapeutic distributions globally, primarily because these products require careful handling, proper storage, as well as trained pharmaceutical staff who understand the complexities of coagulation disorders as well as immune deficiencies.
Recombinant C1-inhibitors are made using new methods to make safe as well as scalable treatments. For example, Pharming Group uses rabbit milk to make Ruconest (conestat alfa), which operates like human protein, avoids blood-borne infection risks, as well as can be produced in large amounts. This method has been successful, earning €47.2 million for Pharming in 2023, and the drug is approved for treating HAE attacks in the EU (2010) and the US (2014).
Other production way include mammalian cell culture, particularly CHO cells, etc., which offer scalability but face challenges in achieving proper glycosylation, as well as plant-based systems, such as transient expression in Nicotiana benthamiana, which are pocket friendly but still showing regulatory hurdles. These diverse platforms collectively advance the development of recombinant C1-inhibitor therapies with improved safety, supply, as well as production flexibility.
|
Current Event |
Description and its Impact |
|
FDA Rare Disease Guidance Update (2025) |
|
|
FDA Approval of Subcutaneous C1-Inhibitor Formulations (2024) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
To learn more about this report, Request Free Sample
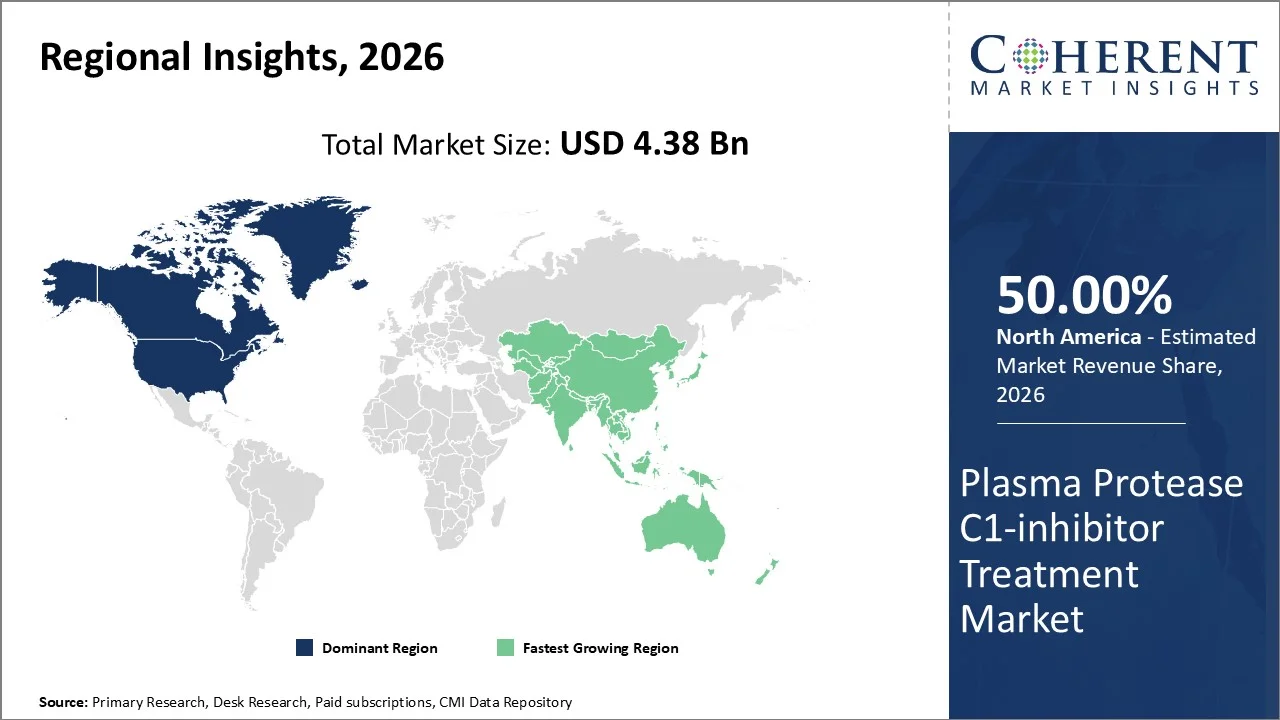
North America account 50% market share in 2026, owing to its bolstering healthcare infrastructure, advanced medical research capabilities, as well as comprehensive regulatory framework that facilitates rapid adoption of specialized treatments like Plasma Protease C1-inhibitor therapy. According to the American Hospital Association, there are 6,100 hospitals in the United States that facilitates specialized treatments.
The region is dominated by the presence of world-class medical institutions such as Mayo Clinic, Johns Hopkins, and Cleveland Clinic, which have established specialized centers for hereditary angioedema and complement system disorders. The National Institutes of Health (NIH) has been instrumental in funding research initiatives focused on complement deficiencies, with recent grants exceeding USD15 million allocated specifically for C1-inhibitor research programs.
The Asia-Pacific region is poised to be the fastest-growing region through 2026-2033, expanding at a CAGR of approximately 7.5%. The region experiencing growth owing to fast expanding healthcare infrastructure as well as increasing diagnostic capabilities across emerging economies. Moreover, significant investments in rare disease diagnosis and treatment facilities, particularly in countries like China, India, and Southeast Asian nations, is another factore responsible for the growth of the market. The Asia-Pacific region, home to 60% of the global population, has the highest number of Persons Living with a rare disease (PLWRD). Similarly, according to a data published in the World Economic Forum, globally, there are around 400 million people living with rare diseases, with around 250 million of them in Asia.
The Asia-Pacific Society for Primary Immunodeficiencies has launched comprehensive training programs for healthcare professionals, advancing recognition as well as diagnosis of hereditary angioedema cases that were previously underdiagnosed.
Government actions are helping patients with C1-inhibitor deficiency. In China, rare disease policies include C1-inhibitor deficiency in national registries, making it easier to find patients and give them treatment. In Japan, the drug agency speeds up approval for C1-inhibitor products, and local companies like Mitsubishi Tanabe Pharma are creating plans to distribute them across the country.
The U.S. contributes the highest share in the Plasma Protease C1-inhibitor Treatment Market in North America owing to its robust healthcare infrastructure, advanced research capabilities, and comprehensive regulatory framework that facilitates faster drug approvals and market access.
The United States Food and Drug Administration (FDA) has been instrumental in approving several C1-inhibitor treatments, including Berinert, Cinryze, as well as Ruconest, providing patients with multiple therapeutic options for hereditary angioedema (HAE) and acquired angioedema management.
Japan contributes the highest share in the Plasma Protease C1-inhibitor Treatment Market in Asia Pacific owing to its highly developed healthcare infrastructure, stringent regulatory framework, as well as established pharmaceutical ecosystem. The country is dominating owing to its advanced diagnostic capabilities and early adoption of specialized treatments for hereditary angioedema (HAE) and other complement-mediated disorders.
Japan's Pharmaceuticals and Medical Devices Agency (PMDA) has maintained a comprehensive approval pathway for plasma-derived therapeutics, facilitating market access for C1-inhibitor products.
Some of the major key players in Plasma Protease C1-inhibitor Treatment Market include, CSL Behring LLC, Takeda Pharmaceutical Company Limited, Ionis Pharmaceuticals, Inc., Pharming Technologies B.V., Centogene AG, BioCryst Pharmaceuticals, and KalVista Pharmaceuticals, Inc.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 4.38 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 9.1% | 2033 Value Projection: | USD 8.06 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
CSL Behring LLC, Takeda Pharmaceutical Company Limited, Ionis Pharmaceuticals, Inc., Pharming Technologies B.V., Centogene AG, BioCryst Pharmaceuticals, and KalVista Pharmaceuticals, Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients