Stargardt Disease Therapeutics Market is estimated to be valued at USD 644.6 Mn in 2026 and is expected to reach USD 4,477.1 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 31.9% from 2026 to 2033.
Analysts’ Views on Global Stargardt Disease Therapeutics Market:
Global stargardt disease therapeutics market is emerging and willwitness growth in the near future due to the involvement of key players who are launching new technology and innovations to the field. These key players are investing heavily in research and development to find effective treatments and therapies for stargardt disease. Growing awareness and support from various organizations and government initiatives is expected to drive growth of the stargardt disease therapeutics market.
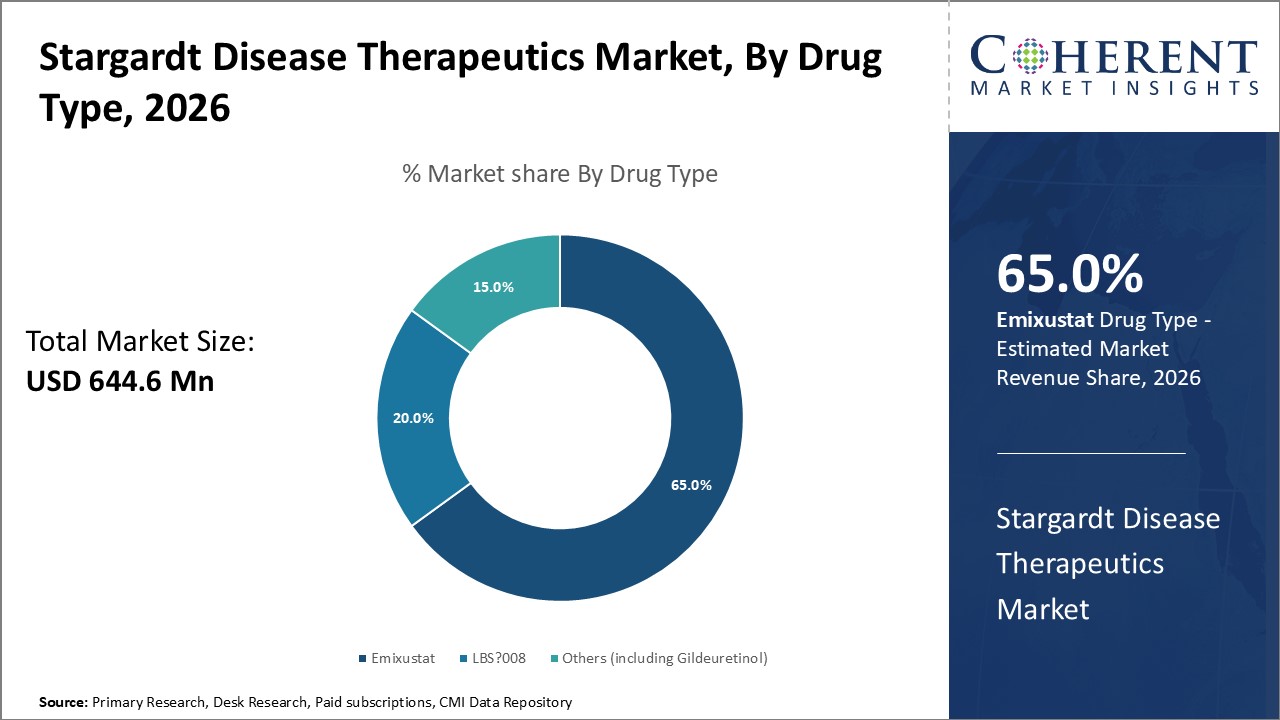
Figure 1. Global Stargardt Disease Therapeutics Market Share (%), By Drug Type, 2026
To learn more about this report, Download Free Sample
Global Stargardt Disease Therapeutics Market– Drivers
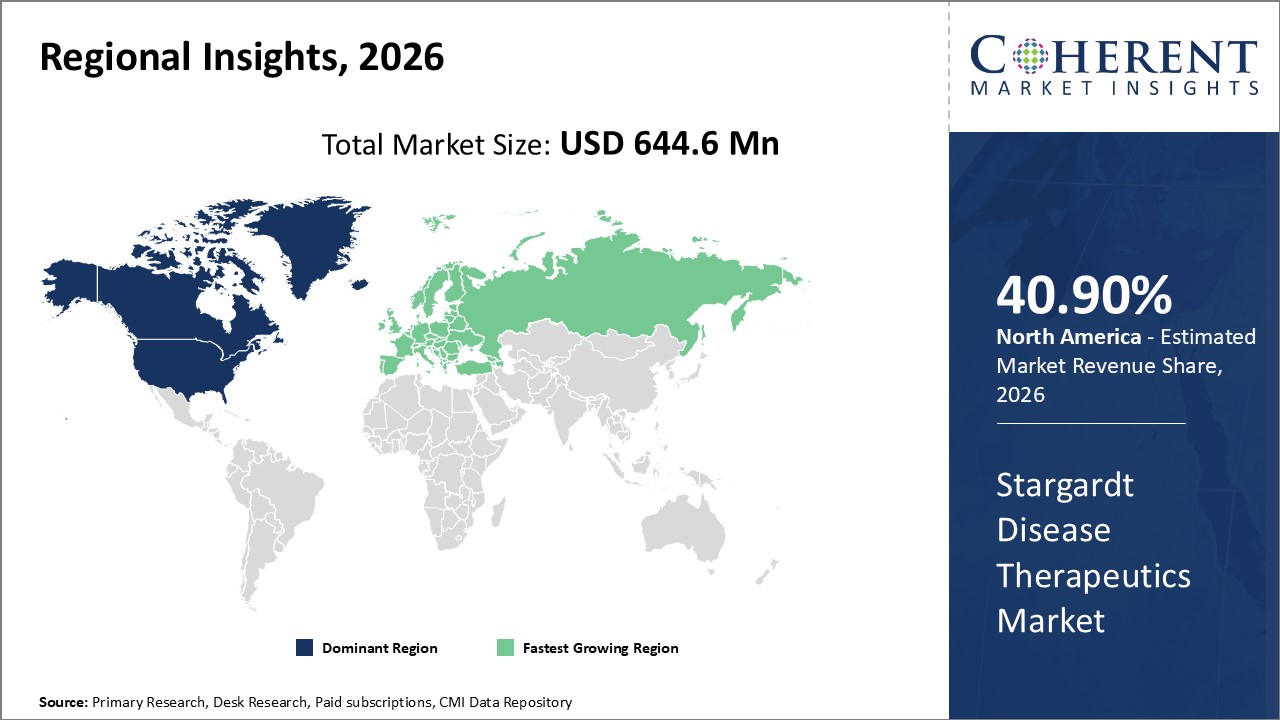
Figure 2. Global Stargardt Disease Therapeutics Market Share(%), By Region, 2026
To learn more about this report, Download Free Sample
Global Stargardt Disease Therapeutics Market- Regional Analysis
Among region, North America is estimated to hold a dominant position in the global stargardt disease therapeutics market over the forecast period. North America is estimated to hold 40.90% of the market share in 2026. Global stargardt disease therapeutics market is expected to witness significant growth in the near future due to introduction of synHSV platform for the treatment of stargardt disease. The synHSV is capable of delivering up to 30x the payload of AAV. It can deliver big genes, genomic genes, and multiple genes, and thus, unlocking the promise of polygenic medicine. The synHSV was launched by REPLAY, a U.S.-based genome writing company that reprogrammes biology by writing and delivering big DNA.
Global Stargardt Disease
Stargardt Disease Therapeutics Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 644.6 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 31.9% | 2033 Value Projection: | USD 4,477.1 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Kubota Pharmaceutical Holdings Co., Ltd., Stargazer Pharmaceuticals Inc., Iveric Bio, Inc., Sanofi S.A., Alkeus Pharmaceuticals Inc., Astellas Pharma Inc., CHABiotech CO., Ltd, ReVision Therapeutics, Inc., Lin BioScience, Inc., Biogen Inc., Hoffmann-La Roche AG, Ocugen, Inc., Ascidian Therapeutics, Nanoscope Therapeutics Inc., Aequus Pharmaceuticals Inc., Fera Pharmaceuticals, LLC, Insmed Incorporated, Belite Bio, Inc., and SpliceBio. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Stargardt Disease Therapeutics Market- Segmentation
Global Stargardt disease therapeutics market report is segmented into by drug type, by age group, by distribution channel, and by region.
Among all the segmentation, the drug type segment is expected to dominate the market over the forecast period due to increasing launch of the drugs, collaboration for the market expansion, and also bringing new technology to meet the unmet needs.
Global Stargardt Disease Therapeutics Market- Cross Sectional Analysis
Rising incidence rate in the North America region is expected to boost demand for stargardt disease therapeutics in North America region. It is estimated prevalence in the U.S. is 10 to 12.5 per 100,000. Cornish et al. estimated its annual incidence in the U.K. is 0.110 to 0.128 per 100 000 individuals.
Global Stargardt Disease Therapeutics Market: Key Developments
In October 2022, ATP, a life sciences venture firm, is the most recent startup to make its debut with a novel approach to genetic editing. With US$ 50 million in funding, U.S.-based Ascidian Therapeutics claims that its RNA “exon editing” approach could match the durability of gene therapy while avoiding some of the risks that come with editing DNA. The platform is designed to correct for mutations in exons,the regions of DNA that contain information needed to make proteins
Global Stargardt Disease Therapeutics Market: Key Trends
Global Stargardt Disease Therapeutics Market: Restraint
Global Stargardt Disease Therapeutics Market - Key Players
Major players operating in the global stargardt disease therapeutics market include Kubota Pharmaceutical Holdings Co., Ltd., Stargazer Pharmaceuticals Inc., Iveric Bio, Inc., Sanofi S.A., Alkeus Pharmaceuticals Inc., Astellas Pharma Inc., CHABiotech CO., Ltd, ReVision Therapeutics, Inc., Lin BioScience, Inc., Biogen Inc., Hoffmann-La Roche AG, Ocugen, Inc., Ascidian Therapeutics, Nanoscope Therapeutics Inc., Aequus Pharmaceuticals Inc., Fera Pharmaceuticals, LLC, Insmed Incorporated, Belite Bio, Inc., and SpliceBio.
Global Stargardt Disease Therapeutics Market– Definition
Stargardt disease (STGD) is the most common childhood recessively inherited macular dystrophy. The condition has a genetic basis due to mutations in the ABCA4 gene, on chromosome 1, that encodes a retinal transported protein. It results from the accumulation of visual cycle kinetics-derived byproducts in the retinal pigmented epithelium (RPE) with secondary photoreceptor dysfunction and death. The research team in Oxford uses a technology called CRISPR to target specific errors in the faulty gene ABCA4, that causes stargardt disease. The team hopes to correct the code, by targeting DNA and RNA so that enough normal ABCA4 protein is made to restore diseased macular cells to health.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients