The global acute otitis media treatment market size is estimated to reach USD 3,360.5 Mn in 2026 and is projected to grow at a compound annual growth rate (CAGR) of 5.0% during the forecast period (2026-2033), surpassing USD 4,728.6 Mn by 2033. This growth can be attributed to rising prevalence of acute otitis media, growing awareness of early diagnosis and treatment options, and improved access to antibiotics and advanced therapeutics.
The global acute otitis media treatment market is expected to grow steadily during the forecast period. This is due to increasing prevalence of acute otitis media, rising incidence of upper respiratory tract infections, growing awareness of early diagnosis and treatment options, and high adoption of antibiotics and advanced therapeutics.
Acute otitis media (AOM) is an infection of the middle ear, the air-filled space behind the eardrum, which commonly causes ear pain, fever, irritability, and difficulty sleeping. It is one of the most common ear infections in children, largely because their Eustachian tube is shorter and more horizontal than in adults, making it easier for pathogens to reach the middle ear and for fluid to accumulate.
The most common bacterial pathogens associated with AOM include Streptococcus pneumoniae, non-typeable Haemophilus influenzae, and Moraxella catarrhalis. Viral infections also frequently contribute to AOM and may precede or coexist with bacterial infection. Rising cases of these infections are expected to create a conducive environment for the growth of acute otitis media treatment during the projection period.
Expansion of telemedicine and digital healthcare is also supporting the growth of the AOM treatment market. Telehealth adoption is enabling remote diagnosis, faster clinical consultations, and easier access to prescriptions through online platforms. This trend is improving treatment accessibility, particularly in rural and underserved regions.
According to Coherent Market Insights’ latest acute otitis media treatment market analysis, antibiotics are set to dominate the industry with a share of nearly 70% in 2026. This dominance can be attributed to antibiotics remaining the first-line treatment for most bacterial cases of acute otitis media, especially in pediatric patients.
Antibiotics are widely available and work effectively against common bacteria such as Streptococcus pneumoniae and Haemophilus influenzae. Their use is also strongly supported by standard treatment guidelines in primary care settings, which helps maintain their high utilization.
For example, a US claims-based study published in Pediatrics (American Academy of Pediatrics journal), analyzing over one million acute otitis media encounters, reported high antibiotic utilization in routine care. Amoxicillin was the most frequently prescribed antibiotic, accounting for approximately half of all prescriptions in real-world datasets.
To learn more about this report, Request Free Sample
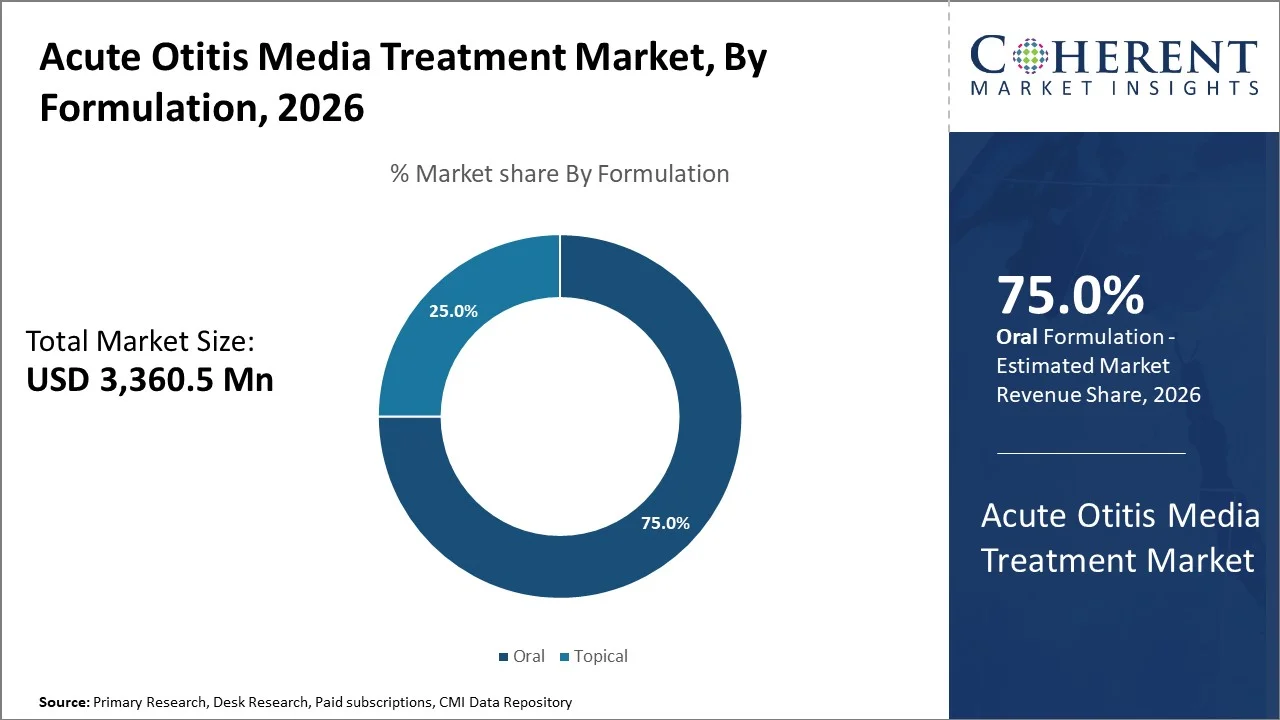
By formulation, oral segment is projected to account for a prominent market share of 75% in 2026. This is mostly due to their non-invasive nature, ease of dosing in pediatric patients, and higher caregiver preference compared to injections or other routes of administration.
Oral antibiotics also support better adherence in outpatient settings, making them the first-line choice in the majority of acute otitis media treatment protocols. As a result, the target segment is expected to lead the global acute otitis media treatment market throughout the forecast period.
Rising Prevalence of Acute Otitis Media (AOM): Increasing incidence of ear infections, especially in children, is providing a strong impetus for the growth of acute otitis media treatment market. Acute otitis media remains one of the most common pediatric infections worldwide, leading to frequent doctor visits and high treatment demand.
For instance, according to studies published in the Likewise, 80% to 90% develop otitis media with effusion before reaching school age. This high AOM burden is expected to fuel demand for acute otitis media treatment drugs during the forecast period.
Increasing Awareness and Early Diagnosis: People across the world are increasingly becoming aware of ear infections, their symptoms, and the availability of treatments. This is leading to earlier diagnosis, faster initiation of treatment, and reduced complications such as hearing loss and mastoiditis. Thus, growing health awareness is expected to create lucrative growth opportunities in the acute otitis media treatment market during the assessment period.
Shift Towards Combination Therapies: Rising adoption of combination and supportive therapies is a key growth-shaping trend in the acute otitis media treatment market. There is a growing shift toward antibiotic and anti-inflammatory combinations, increased use of analgesics for pain management, and symptomatic relief therapies. This transition toward a more holistic treatment approach is expected to boost the acute otitis media treatment market value and growth during the forecast period.
New technologies are improving how acute otitis media (AOM) is diagnosed. Digital otoscopes and AI-based diagnostic tools are helping clinicians assess the eardrum more clearly and support faster diagnosis, especially in telehealth and primary care settings. This enables quicker clinical decisions and improves access to care in regions with limited ENT specialist availability.
A recent example is TytoCare, which received FDA De Novo classification in 2026 for its Tyto Insights™ for ENT Suite. The system uses AI to analyze otoscopy video recordings and detect eardrum bulging, a key clinical sign associated with acute otitis media. This marks a regulatory milestone for AI-powered ENT image analysis and supports more accurate remote ear examinations in virtual care settings. These advancements are expected to improve early diagnosis and support timely treatment decisions, thereby increasing demand for acute otitis media therapeutics.
|
Current Event |
Description and its Impact |
|
High lifetime prevalence of ear infections in children (NIDCD data) |
|
|
Expansion of pediatric pneumococcal vaccination (PCV20 recommendations) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
To learn more about this report, Request Free Sample
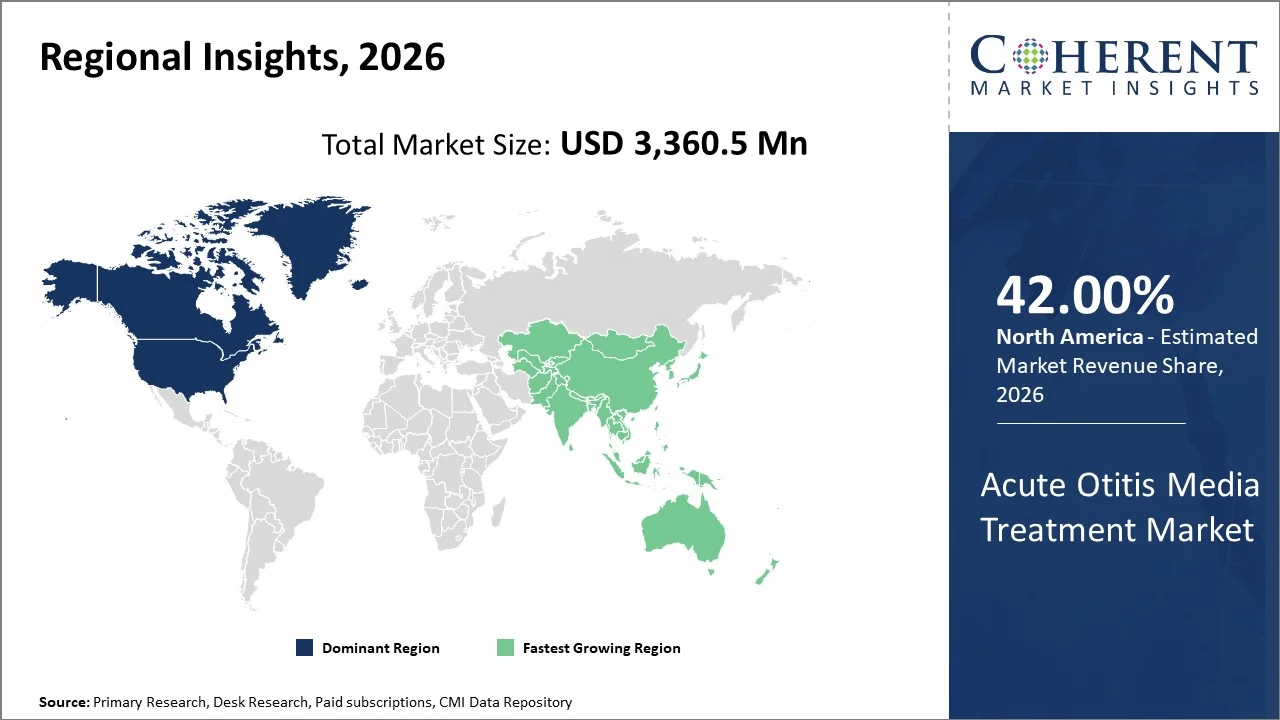
North America is expected to dominate the global acute otitis media treatment market during the forecast period, capturing a share of 42% in 2026. This can be attributed to high disease awareness, strong healthcare access, early diagnosis in children, and presence of leading pharmaceutical companies.
Rising prevalence of AOM in young children is playing a key role in boosting North America acute otitis media treatment market growth. Acute otitis media (OM) is quite common in the United States. Around 60% of children experience it by the age of 3 years. In addition, more than 2.2 million cases of otitis media with effusion (fluid buildup in the middle ear) are diagnosed every year. This is driving demand for acute otitis media treatment drugs, especially antibiotics.
Asia Pacific is emerging as a fast-growing region in the acute otitis media (AOM) treatment market, holding a share of 25% in 2026. This is mostly due to improving healthcare access, rising pediatric population, increasing awareness of ear infections, and increasing demand for antibiotics.
High burden of upper respiratory infections, which often trigger AOM in children, especially in densely populated countries, is positively impacting sales of acute otitis media treatments in the region. Research shows AOM is strongly linked with viral infections like RSV and influenza, which remain common in the region and contribute to recurrent ear infections.
The United States is expected to remain a leading market for acute otitis media (AOM) treatments during the forecast period. This is mostly due to strong healthcare infrastructure, high awareness of ear infections, and frequent diagnosis in pediatric populations. Similarly, the presence of advanced ENT specialists and strong healthcare reimbursement systems also reinforces the country’s position as a mature and highly structured market for acute otitis media treatment.
The acute otitis media treatment market in China is expected to grow rapidly during the assessment period. This can be attributed to rising burden of respiratory tract infections in children, growing health awareness, and rising adoption of antibiotics for acute otitis media. Likewise, improving access to healthcare services, expanding pediatric care infrastructure, and rising healthcare expenditure are further supporting market expansion.
Some of the major players in Acute Otitis Media Treatment Market are Pfizer, Inc., Eli Lilly and Company, Abbott Laboratories, GlaxoSmithKline plc., Bayer AG, Novartis AG, Pediapharm Inc., Sanofi S.A., and Bristol Myers Squibb Company.
Leading acute otitis media treatment companies are adopting various strategies to boost their revenue as well as gain a competitive edge over the industry. These include new product launches and approvals, increasing investments in R&D for novel antibiotics and improved formulations, development of child-friendly dosage forms, collaborations, mergers, and acquisitions.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 3,360.5 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.0% | 2033 Value Projection: | USD 4,728.6 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Pfizer, Inc., Eli Lilly and Company, Abbott Laboratories, GlaxoSmithKline plc., Bayer AG, Novartis AG, Pediapharm Inc., Sanofi S.A., and Bristol Myers Squibb Company. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients